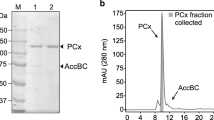
Summary
Alcohol dehydrogenase (EC 1.1.1.1) and pyruvate decarboxylase (EC 4.1.1.1) from Zymomonas mobilis were partially purified and characterized. Alcohol dehydrogenase exhibits a pH optimum at 6.5 and pyruvate decarboxylase a major peak at pH 6.0 and a minor one at pH 4.3. The molecular weights were estimated to be 147,300±14,700 and 219,700±20,400 daltons, respectively. Both enzymes exhibit hyperbolic saturation curves with their respective substrates. Whereas alcohol dehydrogenase was inhibited by ethanol (Ki=6.86×10−4 M) and NAD+ (Ki=1.44×10−4 M), no inhibition was observed with pyruvate decarboxylase at similar concentrations of ethanol. Neither of the enzymes responded to sulphydryl-binding reagents, but differed in their response to a number of divalent metals. The results are compared with those of the respective enzymes from yeast and discussed with a view towards ethanol production limitations by Zymomonas mobilis.
Similar content being viewed by others
References
Arcuri EJ (1982) Continuous ethanol production and cell growth in an immobilized-cell bioreactor employing Zymomonas mobilis. Biotechnol Bioeng 24:595–604
Arcuri EJ, Worden RM, Shumate II SW (1980) Ethanol production by immobilized cells of Zymomonas mobilis. Biotechnol Letts 2:499–504
Biellman JF, Jung MJ (1971) Mechanism of alcohol dehydrogenase from yeast and horse liver. Eur J Biochem 19:130–136
Boiteaux A, Hess B (1970) Allosteric properties of yeast pyruvate decarboxylase. FEBS-Letts 9:293–296
Bränden S, Jöruvall H, Eklund B, Furugren B (1975) Alcohol dehydrogenases. In: ‘The Enzymes’ 11: 104–164
Buhner M, Sund H (1969) Yeast alcohol dehydrogenase-SH groups, disulphide groups, quarternary structure and reactivation by reductive cleavage of disulphide groups. Eur J Biochem 11:73–79
Cromie S, Doelle HW (1980) Relationships between maintenance energy requirement, mineral salts and efficiency of glucose to ethanol conversion by Zymomonas mobilis. Biotechnol Letts. 2:357–362
Cromie S, Doelle HW (1981) Nutritional effects on the kinetics of ethanol production from glucose by Zymomonas mobilis. Eur J Appl Microbiol Biotechnol 11:116–119
Cromie S, Doelle HW (1982) Limitations in substrate utilization efficiency by Zymomonas mobilis. Eur J Appl Microbiol Biotechnol 14:69–73
Doelle HW (1982a) The existence of two separate constitutive enzymes for glucose and fructose in Zymomonas mobilis. Eur J Appl Microbiol Biotechnol 15:20–24
Doelle HW (1982b) Kinetic characteristics and regulatory mechanisms of glucokinase and fructokinase from Zymomonas mobilis. Eur J Appl Microbiol Biotechnol 14:241–246
Doelle HW, Preusser HJ, Rostek H (1982) Electronmicroscopic investigations of Zymomonas mobilis cells grown in low and high glucose concentrations. Eur J Appl Microbiol Biotechnol 16:136–141
Doelle HW, McGregor AN (1983) The effect of high ethanol and CO2 concentrations on the ultrastructure of Zymomonas mobilis. Eur J Appl Microbiol Biotechnol 17:44–48
Gounaris AD, Turkenkopf I, Civerchia LL, Greenlie J (1975) Pyruvate decarboxylase. III. Specificity restrictions for thiamine pyrophosphate in the protein-association step, sub-unit structure. Biochim Biophys Acta 405:492–499
Hayes JE, Velick SF (1954) Yeast alcohol dehydrogenase, molecular weight, coenzyme binding and reaction equilibria. J Biol Chem 207:225–244
Hoppe GK, Hansford GS (1982) Ethanol inhibition of continuous anaerobic yeast growth. Biotechnol Letts 4:39–44
Hubner G, Schellenberger A (1980) Molecular aspects of the function of yeast pyruvate decarboxylase. In: Milduer P, Ries B, (eds) Trends in Enzymology. Enzyme regulation and mechanism of action. Pergamon Press, Oxford, pp 331–339
Jordan F, Kuo DJ, Mouse EU (1978) A pH rate determination of the activity Ph profile of enzymes. Application to yeast pyruvate decarboxylase demonstrating the existence of multiple ionisable groups. Anal Biochem 86:298–302
Ludewig R, Schellenberger A (1974) A new procedure to prepare highly purified and crystallized yeast pyruvate decarboxylase. FEBS-Letts 45:340–343
Lustdorf PU, Megnet R (1968) Multiple forms of alcohol dehydrogenase in Saccharomyces cerevisiae. I. Physiological control of ADH-2 and ADH-4. Arch Biochem 126:933–944
Millar DG, Griffiths-Smith K, Algar E, Scopes RK (1982) Activity and stability of glycolytic enzymes in the presence of ethanol. Biotechnol Letts (in press)
Novak M, Strabaiano P, Morena M, Goma G (1981) Alcoholic fermentation: On the inhibitory effect of ethanol. Biotechnol Bioeng 23:201–211
Rashid N, Rabin BR (1968) Inhibition of yeast alcohol dehydrogenase by alkylating agents. Eur J Biochem 5:147–150
Rogers PL, Lee KJ, Skotincki M, Tribe DE (1982) Ethanol production by Zymomonas mobilis. Adv. Biochem Eng 23:37–84
Schellenberger G (1967) Structure and mechanism of action of the active center of yeast pyruvate decarboxylase. Angew Chem, Intern Edn 6:1024–1035
Sokatch JR (1979) Roles of appendages and surface layers in adaptation of bacteria to their environment. In: Sokatch JR, Ornston LN, (eds) The Bacteria. Academic Press, pp 229–289
Ullrich J (1970) Yeast pyruvate decarboxylase (2-oxoacid carboxy-lyase EC 4.1.1.1), assay of thiamine pyrophosphate. Meth Enzymol 18:109–115
Ullrich J, Wittorf JH, Gubler CJ (1965) Molecular weight and coenzyme content of pyruvate decarboxylase from brewer's yeast. Biochim Biophys Acta 113:595–604
Ullrich J, Donner I (1970) Kinetic evidence for two active sites in cytoplasmic yeast pyruvate decarboxylase. Hoppe-Seyler's Z Physiol Chem 351:1026–1029
Ullrich J, Ostrovsky YM, Eyzaguire J, Holzer H (1971) Thiamine pyrophosphate catalyzed enzymatic decarboxylation of α-axo acids. Vit Horm 28:365–398
Wenger JI, Bernsofky C (1967) Mitochondrial alcohol dehydrogenase from Saccharomyces cerevisiae. Biochem Biophys Acta 227:479–482
Wills C, Kraftofil P, Loudo D, Martin T (1981) Characterization of the two alcohol dehydrogenases of Zymomonas mobilis. Arch Biochem Biophys 210:775–785
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hoppner, T.C., Doelle, H.W. Purification and kinetic characteristics of pyruvate decarboxylase and ethanol dehydrogenase from Zymomonas mobilis in relation to ethanol production. European J. Appl. Microbiol. Biotechnol. 17, 152–157 (1983). https://doi.org/10.1007/BF00505880
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00505880