Abstract
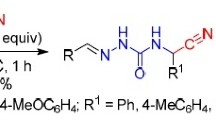
The cyclization of N-alkylated (arylated) α-ureidocarboxylic acids gives a series of 3-mono- and 3,5-disubstituted hydantoins, and nitrosation of the latter yields their nitroso derivatives. The rotational isomerism of 3-(α-naphthyl)hydantoins has been studied by PMR spectroscopy.
Similar content being viewed by others
Literature cited
V. V. Lutsenko, R. A. Blyum, and I. L. Knunyants, Zh. Org. Khim., 7, 1152 (1971).
V. V. Lutsenko, Z. A. Stumbryavichyute, and Z. L. Zhimantene, Zh. Org. Khim., 14, 1203 (1978).
R. S. Baltrushis, Z.-I. G. Beresnyavichyus, I. M. Vizgaitis, and Yu. V. Gatilov, Khim. Geterotsikl. Soedin., No. 12, 1669 (1981).
V. F. Bystrov, Usp. Khim., 41, 512 (1972).
H. Fujiwara and J. M. van der Veen, J. Chem. Soc., Perkin Trans. II, No. 5, 659 (1979).
R. E. Cassady and S. W. Hawkinson, Acta Crystallogr., 38B, 1646 (1982).
L. D. Colebrook, H. G. Giles, A. Granata, S. Icli, and J. R. Fehlner, Can. J. Chem., 51, 3635 (1973).
L. D. Colebrook, S. Icli, and F. H. Hund, Can. J. Chem., 53, 1556 (1975).
W. E. Bentz, L. D. Colebrook, J. R. Fehlner, and A. Rosowsky, Chem. Commun., No. 16, 974 (1970).
V. V. Lutsenko, Zh. Org. Khim., 15, 1973 (1979).
K.-A. V. Klimavichyus, R. A. Blyum, V. I. Shimkyavichene, Ya. V. Valavichene, and V. V. Lutsenko, in: Proceedings of the All-Union Conference on Timely Problems in the Experimental Chemotherapy of Tumors [in Russian], Chernogolovka (1980), p. 43.
H. Aspelund, Acata Acad. Aboensis, Math. Phys., No. 23, 2 (1962).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1404–1406, October,
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1404–1406, October,
Rights and permissions
About this article
Cite this article
Klimavichyus, K.A.V., Lutsenko, V.V., Paulauskaite, G.V. et al. Synthesis and nitrosation of 3- and 3,5-substituted hydantoins. Chem Heterocycl Compd 20, 1160–1162 (1984). https://doi.org/10.1007/BF00503612
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00503612