Abstract
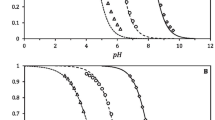
The two most common variants of placental alkaline phosphatase, the F and S variants, were purified to homogeneity and characterized. Their molecular weights were determined by equilibrium ultracentrifugation and sodium dodecylsulfate polyacrylamide gel electrophoresis, which gave almost identical values for the two variants, 118,000 (F) and 119,000 (S). The amino acid compositions of the F and S variants presented here are found to be very similar. Differences between the two variants were found in specific activity (160 U/mg for F and 250 U/mg for S), isoelectric point (IP=4.5 for F and 4.7 for S), sedimentation coefficient (6.5×10−13 sec for F and 6.4×10−13 sec for S). Thus the structural differences observed for these enzyme variants seem to affect both the active site and the protein conformation.
Similar content being viewed by others
References
Beckman, G. (1970). Genetics of human placental phosphatases. Thesis, Umeå.
Beckman, G., Beckman, L., and Cristodoulou, D. (1966). Pregnancy enzymes and placental polymorphism. I. Alkaline phosphatase. Acta Genet. Basel 1659.
Beckman, G., Beckman, L., and Magnusson, S. S. (1972). Placental alkaline phosphatase phenotypes and pre-natal selection. Hum. Hered. 22473.
Beckman, L., Beckman, G., Cristodoulou, D., and Ifekwuningwe, A. (1967). Variations in human placental alkaline phosphatase. Acta Genet. Basel 17406.
Beratis, N., Seegers, W., and Hirschhorn, K. (1970). Properties of placental alkaline phosphatase. I. Molecular size and electrical charge of the various electrophoretic components of the six common phenotypes. Biochem. Genet. 4689.
Bowers, G. N., and McComb, R. B. (1966). A continuous spectrophotometric method for measuring the activity of serum alkaline phosphatase. Clin. Chem. 1270.
Boyer, S. H. (1961). Alkaline phosphatase in human sera and placentae. Science 1341002.
Boyer, S. H. (1963). Human organ alkaline phosphatases: Discrimination by several means including starch gel electrophoresis of antienzyme-enzyme supernatant fluids. Ann. N. Y. Acad. Sci. 103938.
Chervenka, C. H. (1970). Long-column meniscus depletion sedimentation equilibrium technique for the analytical ultracentrifugation. Anal. Biochem. 3424.
Chiandussi, L., Greene, S. F., and Sherlock, S. (1962). Serum alkaline phosphatase fractions in hepato-biliary and bone diseases. Clin. Sci. 22425.
Cinader, B. (1963). Immunochemistry of enzymes. Ann. N. Y. Acad. Sci. 103495.
Cohn, E. J., and Edsall, J. P. (1943). Proteins, Amino Acid and Peptides as Ions and Dipolar Ions, Reinhold, New York, p. 372.
Cunningham, V. R., and Rimer, J.-G. (1963). Isoenzymes of alkaline phosphatase of human serum. Biochem. J. 8950.
Davis, B. J. (1964). Disc electrophoresis II. Method and application to human serum proteins. Ann. N. Y. Acad. Sci. 121404.
Doellgast, G. J., and Fishman, W. H. (1974). Purification of human placental alkaline phosphatase. Biochem. J. 141103.
Fishman, W. H., Green, S., and Inglis, N. R. (1963). L-Phenylalanine: An organ specific, stereospecific inhibitor of human intestinal alkaline phosphatase. Nature 198685.
Ghosh, N. K., and Fishman, W. H. (1968). Purification and properties of molecular-weight variants of human placental alkaline phosphatase. Biochem. J. 108779.
Green, S., Cantor, F., Inglis, N. R., and Fishman, W. H. (1972). Normal serum alkaline phosphatase isoenzymes examined by acrylamide and starch gel electrophoresis and by isoenzyme analysis using organ-specific inhibitors. Am. J. Clin. Pathol. 5752.
Gutman, A. B. (1959). Serum alkaline phosphatase activity in diseases of the skeletal and hepatobiliary systems. Am. J. Med. 27875.
Haije, W. B., and De Jong M. (1963). Isoenzyme patterns of serum alkaline phosphatase in agar-gel electrophoresis and their clinical significance. Clin. Chim. Acta 8620.
Hafkenscheid, J. C. M., and Jansen, A. P. (1975). Standardization of the alkaline phosphatase determination with human placental phosphatase. Clin. Chim. Acta 5963.
Hodson, A. W., Latner, A. L., and Raine, L. (1962). Isoenzymes of alkaline phosphatase. Clin. Chim. Acta 7255.
Kreisher, J. H., Close, V. A., and Fishman, W. H. (1965). Identification by means of L-phenylalanine inhibition of intestinal alkaline phosphatase components separated by starch gel electrophoresis of serum. Clin. Chim. Acta 11122.
Landau, W., and Schlamowitz, M. (1961) Studies of factors related to the differentiation of alkaline phosphatases derived from several tissues. Arch. Biochem. Biophys. 95474.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193265.
Metzger, H., Shapiro, M. B., Mosimann, J. E., and Vinton, J. E. (1968). Assessment of compositional relatedness between proteins. Nature 2191166.
Moore, S., Spackman, D. H., and Stein, W. H. (1958). Chromatography of amino acids on sulfonated polystyrene resins. Anal. Chem. 301185.
Morton, R. K. (1955). The substrate specificity and inhibition of alkaline phosphatases of cow's milk and calf intestinal mucosa. Biochem. J. 61232.
Moss, D. W. (1961). Characterization of tissue alkaline phosphatases and their partial purification by starch-gel electrophoresis. Biochem. J. 81441.
Moss, D. W., and King, E. J. (1962). Properties of alkaline phosphatase fractions separated by starch-gel electrophoresis. Biochem. J. 84192.
Nathanson, L., and Fishman, W. H. (1971). New observations on the Regan isoenzyme of alkaline phosphatase in cancer patients. Cancer 271388.
Nisselbaum, J. A., Schlamowitz, M., and Bodansky, O. (1961). Immunochemical studies of functionally similar enzymes. Ann. N. Y. Acad. Sci. 94970.
Pelichova, H., Kraml, J., Chmelar, M., and Vecerek, B. (1965). Precipitating and binding antibodies against human intestinal alkaline phosphatase. Folia Biochem. 11208.
Posen, S., Neale, F. C., and Clubb, J. S. (1965). Heat inactivation in the study of human alkaline phosphatases. Ann. Intern. Med. 621234.
Robson, E. B., and Harris, H. (1965). Genetics of the alkaline phosphatase polymorphism of the human placenta. Nature 2071257.
Rosenberg, I. N. (1959). Zone electrophoretic studies of serum alkaline phosphatase. J. Clin. Invest. 38630.
Schlamowitz, M. (1958). Immunochemical studies of alkaline phosphatase. Ann. N. Y. Acad. Sci. 75373.
Schlamowitz, M., and Bodansky, O. (1959). Tissue sources of human serum alkaline phosphatase, as determined by immunochemical procedures. J. Biol. Chem. 2341433.
Shields, R., and Burnett, W. (1960). Determination of protein-bound carbohydrate in serum by a modified anthrone method. Anal. Chem. 32885.
Sussman, H. H., Small, P. A., Jr., and Cotlove, E. (1968). Human alkaline phosphatase: Immunochemical identification of organ-specific isoenzymes. J. Biol. Chem. 243160.
Usategui-Gomez, M., Yeager, F., and Fernandez de Castro, A. A. (1973). Sensitive immunochemical method for the determination of the Regan isoenzyme in serum. Cancer Res. 331574.
Usategui-Gomez, M., Yeager, F. M., and Tarbutten, P. (1974a). Purification of human placental alkaline phosphatase by isoelectric focusing. Clin. Chim. Acta 50405.
Usategui-Gomez, M., Yeager, F., and Fernandez de Castro, A. A. (1974b). Regan isoenzyme in normal human sera. Cancer Res. 342544.
Warnock, M. L. (1966). Characterization of tissue and serum alkaline phosphatases. Clin. Chim. Acta 14156.
Weber, K., and Osborn, M. (1969). The reliability of molecular weight determinations by sulfate-polyacrylamide gel electrophoresis. J. Biol. Chem. 2444406.
Author information
Authors and Affiliations
Additional information
This investigation was supported by grants from the Swedish Medical Research Council and the Medical Faculty, University of Umeå.
Rights and permissions
About this article
Cite this article
Holmgren, P.Å., Stigbrand, T. Purification and partial characterization of two genetic variants of placental alkaline phosphatase. Biochem Genet 14, 777–789 (1976). https://doi.org/10.1007/BF00485341
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00485341