Abstract
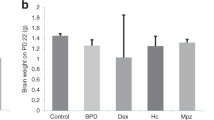
Eighteen infants were entered into a randomized, placebo-controlled trial of dexamethasone therapy for chronic lung disease. Initial ventilation requirements were similar in the two groups, although all infants were in headbox oxygen on entry to the trial. The dexamethasone-treated infants showed a significantly more rapid improvement during the 1st week of treatment, although the overall duration of oxygen therapy was similar in both groups. Cranial ultrasound examination revealed new periventricular abnormalities in three out of the five dexamethasone-treated infants who had previously normal scans, compared with none of four similar placebotreated infants. A large trial, focussing on potential complications, is now needed.
Similar content being viewed by others
References
Avery GB, Fletcher AB, Kaplan M, Brudno DS (1985) Controlled trial of dexamethasone in respirator-dependent infants with bronchopulmonary dysplasia. Pediatrics 75:106–111
Bonta BW, Otero L (1985) Efficacy of dexamethasone in the management of progressive bronchopulmonary dysplasia. Pediatr Res 19:335A
Greenough A, Roberton NRC (1985) Morbidity and survival in neonates ventilated for respiratory distress syndrome. Br Med J 290:597–600
Regev R, DeVries LS, Noble-Jamieson CM, Silverman M (1987) Dexamethasone and increased intracranial echogenicity. Lancet I:632–633
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Noble-Jamieson, C.M., Regev, R. & Silverman, M. Dexamethasone in neonatal chronic lung disease: pulmonary effects and intracranial complications. Eur J Pediatr 148, 365–367 (1989). https://doi.org/10.1007/BF00444135
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00444135