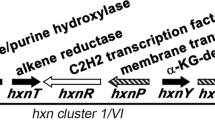
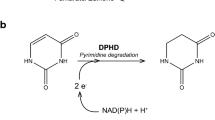
Summary
The complete nucleotide sequence of the Aspergillus nidulans pyruvate kinase gene, including its flanking sequences, is presented. The gene has a 1,578 by coding sequence that encodes a protein of 526 amino acids; the latter is strongly homologous to the pyruvate kinases found in Saccharomyces cerevisiae (66%) and mammals (53%).
The gene is interrupted by seven introns, three of which are in a conserved position compared to those present in the mammalian pyruvate kinase genes sequenced thus far. A fourth intron within the mononucleotide binding fold domain is in a conserved position with respect to the position of an intron within the NAD+ binding region of maize ADH 1.
The transcription start site has been determined; a major site of transcription was found 80 by before the translation initiation codon. The promoter region of the A. nidulans pyruvate kinase gene contains no direct homologies with the TATA or CCAAT sequences in the expected region (30–70 bp) before the transcription initiation site. However, extended CT-enriched regions are found in the promotor region, similar to what has been observed in genes that are highly expressed in Saccharomyces cerevisiae and filamentous fungi.
Similar content being viewed by others
Abbreviations
- bp :
-
base pair(s)
- NAD :
-
nicotinamide dinucleotide
- ORF :
-
open reading frame
References
Bezares G, Eyzaguirre J, Hinrichs MV, Heinrikson RL, Reardon I, Kemp RG, Latshaw SP, Bazaes S (1987) Arch Biochem Biophys 253:133–137
Birnstiel ML, Busslinger M, Strub K (1985) Cell 41:349–359
Bornman L, Röschlau, Hess B (1972) Hoppe-Seyler's Z Physiol Chem 353:696
Burke RL, Tekamp-Olsen P, Najarian R (1983) J Biol Chem 258:2193–2201
Buxton FP,, Gwynne DI, Garven S, Sibley S, Davies RW (1987) Gene 60:255–266
Calzone FJ, Britten RJ, Davidson EH (1987) In: Berger SL, Kimmel AR (eds) Methods in enzymology, vol 152. Academic Press, New York, pp 611–632
Clements JM, Roberts CF (1986) Gene 44:97–105
Cognet M, Lone YC, Vaulont S, Kahn A, Marie J (1987) J Mol Biol 196:11–25
Corrick CM, Twomey AP, Hynes MJ (1987) Gene 53:63–71
Cottam GL, Hollenberg PF, Coon M (1969) J Biol Chem 244: 1481–1486
de Graaff LH, van den Broek HWJ, Visser J (1988) Curr Genet 13:315–321
Dennis ES, Gerlach WL, Pryor AJ, Bennetzen JL, Inglis A, Llewellyn D, Sachs MM, Ferl RJ, Peacock WJ (1984) Nucleic Acid Res 12:3983–4000
Dobson MJ, Tuite MF, Roberts NA, Kingsman AJ, Kingsman SM (1982) Nucleic Acid Res 10:2625–2637
Engström L, Ekman P, Humble E, Zetterqvist O (1987) In: Boyer PD, Krebs EG (eds) The enzymes, vol XVIII. Academic Press, New York, pp 47–75
Fothergill-Gillmore (1986) TIBS 11:47–51
Friedenthal M, Roselino E, Passerson S (1973) Eur J Biochem 35:148–158
Gilbert W (1986) Science 228:823–824
Gwynne DI, Buxton FP, Sibley S, Davies RW, Lockington RA, Scazzocchio C, Sealy-Lewis HM (1987) Gene 51:205–216
Hamer JE, Timberlake WE (1987) Mol Cell Biol 7:2352–2359
Humble E (1980) Biochem Biophys Acta 626:179–187
Inoue H, Noguchi T, Tanaka T (1986) Eur J Biochem 154: 465–469
Johnson SC, Bailey T, Becker RR, Cardenas JM (1979) Biochem Biophys Res Commun 90:525–530
Kapoor M (1976) Int J Biochem 7:439–443
Kester H, Uitzetter JHAA, de Graaff LH, Visser J (1988) Can J Biochem 34:1154–1158
Kozak M (1984) Nucleic Acid Res 12:857–872
Lonberg N, Gilbert W (1983) Proc Natl Acad Sci USA 80:3661–3665
Lonberg N, Gilbert W (1985) Cell 40:81–90
Lone YC, Simon M, Kahn A, Marie J (1986) FEBS Lett 195: 97–100
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY
Maniatis T, Goodburn S, Fischer JA (1987) Science 236:1237–1244
Marie J, Simon MP, Dreyfus JC, Kahn A (1981) Nature 292: 70–72
McKnight GL, Kato H, Upshall A, Parker MD, Saari G, O'Hara PJ (1985) EMBO J 4:2093–2099
McKnight GL, O'Hara PJ, Parker ML (1986) Cell 46:143–147
McLauchlan J, Gaffney D, Whitton JL, Clements JB (1985) Nucleic Acid Res 13:1347–1368
Meixner-Monori B, Kubicek CP, Rohr M (1984) Can J Microbiol 4:16–22
Messing J (1983) Methods Enzymol 101C:20–78
Meyers RM, Tilly K, Maniatis T (1986) Science 232:613–618
Muirhead H, Clayden DA, Barford D, Lorimer CG, Fothergill-Gilmore LA, Schiltz E, Schmitt W (1986) EMBO J 5:475–481
Mullaney EJ, Hamer JE, Roberti KA, Yelton MM, Timberlake WE (1985) Mol Gen Genet 199:37–45
Noguchi T, Inoue H, Tanaka T (1986) J Biol Chem 261:13807–13812
Noguchi T, Yamada K, Inoue H, Matsuda T, Tanaka T (1987) J Biol Chem 262:14366–14371
Norrander J, Kempe T, Messing J (1983) Gene 26:101–106
Pickett M, Gwynne DI, Buxton FP, Elliott R, Davies RW, Lockington RA, Scazzocchio C, Sealy-Lewis HM (1987) Gene 51:217–226
Pontecorvo G, Roper JA, Hemmons LJ, MacDonald KD, Bufton AWJ (1953) Adv Genet 5:141–238
Proudfoot NJ, Brownlee GG (1976) Nature 263:211–214
Rambosek J, Leach J (1987) CRC Crit Rev Biotechnol 6:357–393
Sanger F, Nickelen S, Coulson AR (1977) Proc Natl Acad Sci USA 74:5463–5467
Stuart DI, Levine M, Muirhead H, Stammers DK (1979) J Mol Biol 134:109–142
Tani K, Fujii H, Nagata S, Miwa S (1988) Proc Natl Acad Sci USA 85:1892–1795
Uitzetter JHAA (1982) PhD thesis. Agricultural University of Wageningen, The Netherlands
Upshall A, Gilbert T, Saari G, O'Hara PJ, Weglenski P, Berse B, Miller K, Timberlake WE (1986) Mol Gen Genet 204: 349–354
Valentini G, Iadarola P, Somani BL, Malcovati M (1979) Biochem Biophys Acta 570:248–258
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Graaff, L., Visser, J. Structure of the Aspergillus nidulans pyruvate kinase gene. Curr Genet 14, 553–560 (1988). https://doi.org/10.1007/BF00434080
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00434080