Abstract
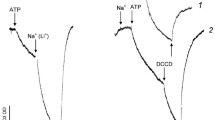
Acidification of weakly buffered suspensions of the cyanobacteria Anacystis nidulans, Nostoc sp. strain MAC, Dermocarpa sp. and Anabaena variabilis was observed after the application of oxygen pulses to anaerobic cells. The acidification was caused by proton extrusion from the oxygen pulsed cells since it was eliminated by the uncoupler (H+ ionophore) carbonyl cyanide m-chlorophenylhydrazone. Results with the inhibitors dicyclohexylcarbodiimide or 7-chloro-4-nitrobenz-2-oxa-1,3-diazole, orthovanadate and cyanide indicated the association of various fractions of the observed proton extrusion with different activities of the cell membrane, viz. a H+-translocating reversible F0F1-ATPase, a unidirectional H+-translocating ATP hydrolase, and a respiratory electron transport system, respectively. Further parameters investigated were the pH dependence and the H+/O stoichiometry of the H+ extrusion from oxygen pulsed cyanobacteria. H+/O ratios at neutral pH were between 4 (Anacystis nidulans) and 0.3 (Dermocarpa) with uninhibited, actively phosphorylating cells and between 2 (Anacystis nidulans) and 0.4 (Dermocarpa) with ATPase-inhibited (ATP-depleted) cells, respectively. It is significant that with all four cyanobacteria tested a major fraction of the observed H+ ejection remained unaffected by ATPase inhibitors even at concentration which completely abolished all oxidative phosphorylation. Vanadate had a major effect on the H+ extrusion from Anabaena only. From this it is concluded that in the cyanobacterial species investigated part of the H+ extrusion from oxygen pulsed cells is directly linked to some H+-translocating respiratory electron transport chain present in the cell membrane.
Similar content being viewed by others
Abbreviations
- CCCP:
-
carbonyl cyanide m-chlorophenylhydrazone
- DCCD:
-
N, N′-dicyclohexylcarbodiimide
- DCMU:
-
N-(3,4-dichlorophenyl-)N′,N′-dimethylurea
- NBD:
-
7-chloro-4-nitrobenzoxa-1,3-diazole
- TPP+ :
-
tetraphenylphosphonium
- Mes:
-
2-(N-morpholino)ethanesulfonic acid
- Pipes:
-
piperazine-N,N′-bis-(2-ethanesulfonic acid)
- Hepes:
-
N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid
- Taps:
-
tris (hydroxymethyl)-methyl-aminopropanesulfonic acid
- Ches:
-
2-(N-cyclohexylamino)-ethanesulfonic acid
- Caps:
-
3-cyclohexylamino)-1-propanesulfonic acid; according to most textbooks (e.g. Nicholls 1982) the terms ”proton electrochemical potential” (\(\Delta \tilde \mu _{{\text{H}}^{\text{ + }} } \)) and “protonmotive force” (pmf, Δp), both of which equivalently describe the energetic state of energy-transducing membranes, were used synonymously and expressed in mV units throughout this article (however, cf. Lowe and Jones 1984)
References
Bowyer JW, Skerman VBD (1968) Production of axenic cultures of soil-borne and endophytic blue-green algae. J Gen Microbiol 54:299–306
Deters DW, Racker E, Nelson N, Nelson H (1975) Partial resolution of the enzymes catalyzing photophosphorylation. XV. Approaches to the active site of coupling factor I. J Biol Chem 250:1041–1047
Goffeau A, Slayman CW (1981) The proton-translocating ATPase of the fungal plasma membrane. Biochim Biophys Acta 639:197–223
Harold FM (1977) Ion currents and physiological functions in microorganisms. Ann Rev Microbiol 31:181–203
Heldt HW, Werdan K, Milovancev M, Geller G (1973) Alkalinization of the chloroplast stroma caused by light dependent proton flux into the thylakoid space. Biochim Biophys Acta 314:224–241
Holowka DA, Hammes GG (1977) Chemical modification and fluorescence studies of chloroplast coupling factor. Biochemistry 16:5538–5545
Kratz WA, Myers J (1955) Nutrition and growth of several bluegreen algae. Am J Bot 42:282–287
Lockau W, Pfeffer S (1982) ATP-dependent calcium transport in membrane vesicles of the cyanobacterium Anabaena variabilis. Biochim Biophys Acta 733:124–132
Lowe AG, Jones MN (1984) Proton motive force—what price Δp? TIBS 9:11–12
Mitchell P (1968) Chemiosmotic coupling and energy transduction. Glynn Research, Bodmin, England
Muchl R, Peschek GA (1983) pH-jump-induced phosphorylation of ADP in the cyanobacterium Anacystis nidulans. FEBS Lett 164:116–120
Muchl R, Peschek GA (1984) Valinomycin pulse-induced phosphorylation of ADP in dark anaerobic cells of the cyanobacterium Anacystis nidulans. Curr Microbiol (in press)
Nicholls D (1982) Bioenergetics: An introduction to chemiosmotic theory. Academic Press, New York
Nitschmann WH (1982) Oxidative Phosphorylierung bei Blaualgen (Cyanobakterien). Ph. D. Thesis, University of Vienna
Nitschmann WH, Peschek GA (1982) Oxidative phosphorylation in intact cyanobacteria. FEBS Lett 139:77–80
Nitschmann WH, Peschek GA (1984) Vanadate and dicyclohexylcarbodiimide insensitive proton extrusion from oxygen pulsed cells of the cyanobacterium Anacystis nidulans. Biochem Biophys Res Commun 123:358–364
Nitschmann WH, Schmetterer G, Muchl R, Peschek GA (1982) Active sodium extrusion reduces net efficiencies of oxidative phosphorylation in the strictly photoautotrophic cyanobacterium Anacystis nidulans. Biochim Biophys Acta 682:293–296
Omata T, Murata N (1984) Cytochromes and prenylquinones in preparations of cytoplasmic and thylakoid membranes from the cyanobacterium (blue-green alga) Anacystis nidulans. Biochim Biophys Acta 766:385–402
Peschek GA (1979) Aerobic hydrogenase activity in Anacystis nidulans: The oxyhydrogen reaction. Biochim Biophys Acta 548:203–215
Peschek GA (1983) Proton pump coupled to cytochrome c oxidase in the cyanobacterium Anacystis nidulans. J Bacteriol 153:539–542
Peschek GA (1984a) Structure and function of respiratory membranes in cyanobacteria. Subcell Biochem 10:85–191
Peschek GA (1984b) Characterization of the proton-translocating cytochrome c oxidase activity in the plasma membrane of intact Anacystis nidulans spheroplasts. Plant Pysiology 75:968–973
Peschek GA, Schmetterer G, Muchl R, Nitschmann WH, Riedler M (1984) Topography and function of cytochrome oxidase in a cyanobacterium. In: Sybesma C (ed) Advances in photosynthesis research, vol III. Martinus Nijhoff & Dr. W. Junk Publishers, The Hague, The Netherlands, pp 335–342
Ramos S, Schuldiner S, Kaback HR (1979) The use of flow dialysis for determination of ΔpH and active transport. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 55, Biomembranes (S. Fleischer, L. Packer, eds). Academic Press, New York, pp 680–688
Rottenberg H (1979) The measurement of membrane potential and ΔpH in cells, organelles and vesicles. In: Colovick SP, Kaplan NO (eds) Methods in enzymology vol 55 Biomembranes (S. Fleischer, L. Packer, eds): 547–569 Academic Press, New York
Scherer S, Böger P (1984) Vanadate-sensitive proton efflux by filamentous cyanobacteria. FEMS Microbiol Letters 22:215–218
Scherer S, Stürzl E, Böger P (1984) Oxygen-dependent proton efflux in cyanobacteria (blue-green algae). J Bacteriol 158:609–614
Schmetterer G, Peschek GA, Sleytr UB (1983) Thylakoid degradation during photooxidative bleaching of the cyanobacterium Anacystis nidulans. Protoplasma 115:202–207
Scholes P, Mitchell P, Myole J (1969) The polarity of proton translocation in some photosynthetic micro-organisms. Eur J Biochem 8:450–454
Sebald W, Machleidt W, Wachter E (1980) N, N′-Dicyclohexylcarbodiimide binds specifically to a single glutamyl residue of the proteolipid subunit of the mitochondrial adenosine triphosphatase from Neurospora crassa and Saccharomyces cerevisiae. Proc Natl Acad Sci USA 77:785–789
Spanswick RM (1981) Electrogenic ion pumps. Ann Rev Plant Physiol 32:267–289
Stanier RY, Kunisawa R, Mandel M, Cohen-Bazire G (1971) Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol Rev 35:171–205
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. G. Drews on the occasion of his 60th birthday
Rights and permissions
About this article
Cite this article
Nitschmann, W.H., Peschek, G.A. Modes of proton translocation across the cell membrane of respiring cyanobacteria. Arch. Microbiol. 141, 330–336 (1985). https://doi.org/10.1007/BF00428845
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00428845