Summary
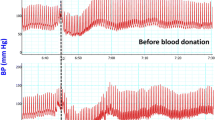
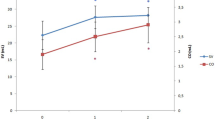
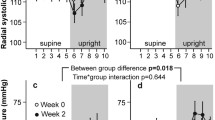
Six healthy males were exposed to 20 mm Hg lower body negative pressure (LBNP) for 8 min followed by 40 mm Hg LBNP for 8 min. Naloxone (0.1 mg·kg−1) was injected intravenously during a 1 h resting period after which the LBNP protocol was repeated. Systolic, mean, and diastolic arterial blood pressures (SAP, MAP, DAP), and central venous pressure (CVP) were obtained using indwelling catheters. Cardiac output (CO), forearm blood flow (FBF), heart rate (HR), left ventricular ejection time (LVET), and electromechanical systole (EMS) were measured non-invasively. Pulse pressure (PP), stroke volume (SV), total peripheral resistance (TPR), forearm vascular resistance (FVR), systolic ejection rate (SER), pre-ejection period (PEP), PEP/LVET and indices for the systolic time intervals (LVETI, EMSI, PEPI) were calculated. During the second LBNP exposure, only two parameters differed from the pre-injection values: DAP at LBNP=40 mm Hg increased from 60.0±4.8 mm Hg to 64.8±4.1mm Hg (N=4, p<0.02) and LVETI at LBNP=20 mm Hg increased from 384.4±5.2 ms to 396.8±6.2 ms (N=6, p<0.02). In connection with the injection, SAP increased from 128.5±4.2 mm Hg to 134.3±5.4 mm Hg (N=6, p<0.025), PP from 56.5+-2.8 mm Hg to 62.7±3.5 mm Hg (N=6, p<0.01), HR from 54.0±3.1min−1 to 59.2±4.1 min−1 (N=6, p<0.01), and LVETI from 407.0±5.6 ms to 413.1±6.0 ms (N=6, p<0.02). This study suggests that endorphins do not have a significant action on the cardiovascular system in the compensated stage of hypovolaemic shock in humans. We found, however, weak evidence that naloxone increases SAP, HR, and LVETI during rest.
Similar content being viewed by others
References
Bonde-Petersen F, Norsk P, Suzuki Y (1980) A comparison between freon and acetylene rebreathing for measuring cardiac output. Aviat Space Environ Med 51:1214–1221
Cohen MR, Cohen RM, Pickar D, Murphy DL, Bunney WE (1982) Physiological effects of high dose naloxone administration to normal adults. Life Sci 30:2025–2031
Dohn K, Gravenhorst JS, Jarløv NV (1956) Volume recorder usable during functional states. Rep Steno Memo Hosp 6:141–168
Faden AI, Holaday JW (1979) Opiate antagonists: A role in the treatment of hypovolemic shock. Science 205:317–318
Faden AI, Holaday JW (1980) Naloxone treatment of endotoxin shock: Stereospecificity of physiologic and pharmacologic effects in the rat. J. Pharmacol Exp Ther 212:441–447
Fuenmayor N, Cubeddu L (1986) Cardiovascular and endocrine effects of naloxone compared in normotensive and hypertensive patients. Eur J Pharmacol 126:189–197
Graboys TB, Forlini FJ, Michaelson ED (1974) Systolic time intervals during lower body negative pressure. J Appl Physiol 37:329–332
Groeger JS, Carlon GC, Howland WS (1983) Naloxone in septic shock. Crit Care Med 11:650–654
Guillemin R, Vargo T, Rossier J, Minick S, Ling N, Rivier C, Vale W, Bloom F (1977) Beta-endorphin and adrenocorticotropin are secreted concomitantly by the pituitary gland. Science 197:1367–1369
Gurll NJ, Reynolds DG, Vargish T, Lechner R (1982a) Naloxone without transfusion prolongs survival and enhances cardiovascular function in hypovolemic shock. J Pharmacol Exp Ther 220:621–624
Gurll NJ, Reynolds DG, Vargish T, Lechner R (1982b) Naltrexone improves survival rate and cardiovascular function in canine hemorrhagic shock. J Pharmacol Exp Ther 220:625–628
Holaday JW, Faden AI (1978) Naloxone reversal of endotoxin hypotension suggests role of endorphins in shock. Nature 275:450–451
Hughes GH, Porter RS, Harker CA, Marx RS (1983a) Naloxone therapy in septic shock. Clin Res 31:258A
Hughes GS, Porter RS, Marx R, Harker CC (1983b) Naloxone and septic shock. Ann Int Med 98:559
Lemaire I, Tseng R, Lemaire S (1978) Systemic administration of beta-endorphin: Potent hypotensive effect involving a serotonergic pathway. Proc Natl Acad Sci 75:6240–6242
Lewis RP, Rittgers SB, Forester WF, Boudoulas H (1977) A critical review of the systolic time intervals. Circulation 56:146–158
Mains RE, Eipper BA, Ling N (1977) Common precursor to corticotropins and endorphins. Proc Natl Acad Sci 74:3014–3018
Peters WP, Johnson MW, Friedman PA, Mitch WE (1981) Pressor effect of naloxone in septic shock. Lancet I:529–532
Reynolds DG, Gurll NJ, Vargish T, Lechner RB, Faden AI, Holaday JW (1980) Blockade of opiate receptors with naloxone improves survival and cardiac performance in canine endotoxic shock. Circ Shock 7:39–48
Rossier J, French ED, Rivier C, Ling N, Guillemin R, Bloom FE (1977) Foot-shock induced stress increases beta-endorphin levels in blood but not in brain. Nature 270:618–620
Salerno TA, Milne B, Jhamandas KH (1981) Hemodynamic effects of naloxone in hemorrhagic shock in pigs. Surg Gynecol Obst 152:773–776
Shatney CH, Cohen RM, Cohen MR, Imagawa DK (1985) Endogenous opioid activity in clinical hemorrhagic shock. Surg Gynecol Obst 160:547–551
Vargish T, Reynolds DG, Gurll NJ, Lechner RB, Holaday JW, Faden AI (1980) Naloxone reversal of hypovolemic shock in dogs. Circ Shock 7:31–38
Weissglas IS, Hinchey EJ, Chiu RCJ (1982) Naloxone and methylprednisolone in the treatment of experimental septic shock. J Surg Res 33:131–135
Weissler AM, Harris WS, Schoenfeld CD (1968) Systolic time intervals in heart failure in man. Circulation 37:149–159
Wiesen M, Liotta AS, Krieger DT (1980) Basal and stimulated release of immunoreactive ACTH, beta-lipotropin and beta-endorphin from human anterior pituitary cells in vitro. Clin Res 28:270A
Wolthuis RA, Bergman SA, Nicogossian AE (1974) Physiological effects of locally applied reduced pressure in man. Physiol Rev 54:566–595
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Foldager, N., Bonde-Petersen, F. Human cardiovascular reactions to simulated hypovolaemia, modified by the opiate antagonist naloxone. Europ. J. Appl. Physiol. 57, 507–513 (1988). https://doi.org/10.1007/BF00418000
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00418000