Abstract
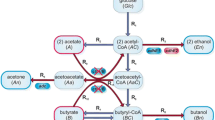
Clostridium acetobutylicum was unable to keep a constant pH inside the cells when grown on a phosphatelimited synthetic medium which allowed production of organic acids in a first phase and of solvents in a second phase. At external pH values between 5.9 and 4.3, the cells kept a constant ΔpH of 0.9 to 1.3. A similar ΔpH was measured in continuous culture under solventproducing conditions. The ΔpH was abolished by protonovorous uncouplers, such as tetrachlorosalicylanilide (TCS) or carbonyl-p-trifluormethoxyphenylhydrazone (FCCP). n-Butanol at concentration of 150 mM and above led also to a complete abolition of the pH gradient.
The internal pH stayed above 5.5 in cultures that shifted from acid to solvent formation. It is concluded that this is a prerequisite for the shift. The possible function of high internal concentrations of butyrate, butyryl phosphate and butyryl coenzyme A in the triggering mechanisms of the shift is discussed.
Similar content being viewed by others
Abbreviations
- TCS:
-
Tetrachlorosalicylanilide
- FCCP:
-
carbonyl-p-trifluormethyoxyphenylhydrazone
References
Andersch W, Bahl H, Gottschalk G (1983) Level of enzymes involved in acetate, butyrate, acetone and butanol formation by Clostridium acetobutylicum. Eur J Microbiol Biotechnol 18:327–332
Bahl H, Gottschalk G (1984) Parameters affecting solvent production by Clostridium acetobutylicum in continuous culture. Biotechnol Bioeng Symp 14:215–223
Bahl H, Andersch W, Gottschalk G (1982a) Continuous production of acetone and butanol by Clostridium acetobutylicum in a twostage phosphate limited chemostat. Eur J Appl Microbiol Biotechnol 15:201–205
Bahl H, Andersch W, Braun K, Gottschalk G (1982b) Effect of pH and butyrate concentration on the production of acetone and butanol by Clostridium acetobutylicum grown in continuous culture. Eur J Appl Microbiol Biotechnol 14:17–20
Bakker EP, Rottenberg H, Caplan SR (1976) An estimation of the light-induced electrochemical potential difference of protons across the membrane of Halobacterium halobium. Biochim Biophys Acta 440:557–572
Baronofsky JJ, Schreurs WJA, Kashket ER (1984) Uncoupling by acetic acid limits growth of and acetogenesis by Clostridium thermoaceticum. Appl Environ Microbiol 48:1134–1139
George HA, Chen J-S (1983) Acidic conditions are not obligatory for onset of butanol formation by Clostridium beijerinckii (synonym. C. butylicum). Appl Environ Microbiol 46:321–327
Gottschal JC, Morris JG (1981) The induction of acetone and butanol production in cultures of Clostridium acetobutylicum by elevated concentrations of acetate and butyrate. FEMS Microbiol Lett 12:385–389
Gottwald M, Hippe H, Gottschalk G (1984) Formation of n-butanol from d-glucose by strains of the “Clostridium tetanomorphum” group. Appl Environ Microbiol 48:573–576
Harold FM, Pavlasova E, Baarda JR (1970) A transmembrane pH-gradient in Streptococcus faecalis: origin and dissipation by proton conductors and N′,N′-Dicyclohexylcarbodiimide. Biochim Biophys Acta 196:235–244
Hartmanis M, Gatenbeck S (1984) Intermediary metabolism in Clostridium acetobutylicum: Level of enzymes involved in the formation of acetate and butyrate. Appl Environ Microbiol 47:1277–1283
Holt R, Stephens G, Morris G (1984) Production of solvents by Clostridium acetobutylicum cultures maintained at neutral pH. Appl Environ Microbiol 48:1166–1170
Kasbket E, Blanchard A Metzger W (1980) Proton motive force during growth of Streptococcus lactis cells. J Bacteriol 143:128–134
Kell DB, Peck MW, Rodger G, Morris JG (1981) On the permeability to weak acids and bases of the cytoplasmic membrane of Clostridium pasteurianum. Biochem Biophys Res Commun 99:81–88
Kobayashi H, Murakami N, Unemoto T (1982) Regulation of the cytoplasmic pH in Streptococcus faecalis. J Biol Chem 257:13246–13252
La Riviere J (1958) On the microbial metabolism of the tartaric acid isomers. Dissertation, Univ. Delft
Long S, Jones D, Woods D (1984) Initiation of solvent production, clostridial stage and endospore formation in Clostridium acetobutylicum P262. Apps Microbiol Biotechnol 20:256–261
Manteufel AJ (1940) Spore formation among Bacteria. II. The conditions of sporulation among acetone-butyl-alcohol bacteria. Microbiologiya J 105-107
Menzel U, Gottschalk G (1985) The internal pH of Acetobacterium wieringae und Acetobacter aceti during growth und production of acetic acid. Arch Microbiol (accompanying paper)
Monot F, Engasser J-M, Petitdemange H (1983) Regulation of acetone butanol production in batch and continuous cultures of Clostridium acetobutylicum. Biotechnol Bioeng Symp 13:207–216
Padan E, Zilberstein D, Schuldiner S (1981) The proton electrochemical gradient in Escherichia coli cells. Eur J Biochem 63:533–541
Prescott SC, Dunn CG (1959) The acetone-butanol fermentation. In: Industrial Microbiology, 3rd edn. McGraw Hill Book Co. New York, pp 250–284
Riebeling V, Thauer R, Jungermann K (1975) The internal-alkaline pH gradient, sensitive to uncoupler and ATPase inhibitor, in growing Clostridium pasteurianum. Eur J Biochem 55:445–453
Rogers P (1985) Genetics and biochemistry of Clostridium relevant to development of fermentation processes. In: Laskin AJ (ed) Advances in Appl Microbiol, vol 31. Academic Press, Inc., London (in press)
Schmidt K, Liaaen-Jensen S, Schlegel HG (1963) Die Carotinoide der Thiorhodaceae. Arch Mikrobiol 46:117–126
Vollherbst-Schneck K, Sands J, Montenecourt B (1984) Effect of butanol on lipid composition and fluidity of Clostridium acetobutylicum ATCC 824. Appl Environ Microbiol 47:193–194
Zeikus JG (1980) Chemical and fuel production by anaerobic bacteria. Annu Rev Microbiol 34:423–456
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gottwald, M., Gottschalk, G. The internal pH of Clostridium acetobutylicum and its effect on the shift from acid to solvent formation. Arch. Microbiol. 143, 42–46 (1985). https://doi.org/10.1007/BF00414766
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00414766