Abstract
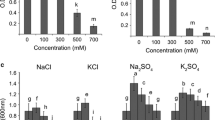
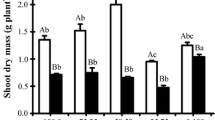
Rhizobium Sp. UMKL 20 responded to increased sodium chloride concentration in the medium by elevating the intracellular concentrations of K+ and glutamate. Increases in K+ occurred in a time course synchronous with glutamate. Addition of the uncoupler 2,4-dinitrophenol, significantly reduced K+ uptake but had little effect on glutamate accumulation. New protein synthesis did not appear to be required for the stimulation of K+ uptake by NaCl. Assays of enzymes involved in glutamate synthesis showed that under salt-stress conditions, increased activities of glutamine synthetase and glutamate synthase were detected, indicating that the GS/GOGAT pathway is the major pathway for increasing intracellular glutamate concentration.
Similar content being viewed by others
References
Bakker EP, Harold RM (1980) Energy coupling to potassium transport in Streptococcus faecalis: interplay of ATP and the proton motive force. J Biol Chem 255:433–440
Balasubramaniam V, Sinha SK (1976) Effects of salt stress on growth, nodulation and nitrogen fixation in cowpeas and mung beans. Physiol Plant 36:197–200
Bernt E, Bergmeyer HU (1974) l-Glutamate: UV assay with GDH and NAD. Meth Enzy Anal IV:1704–1708
Brown AD (1976) Microbial water stress. Bacteriol Rev 40:803–846
Brown CM, Dilworth MJ (1975) Ammonium assimilation by Rhizobium cultures and bacteriods. J Gen Microbiol 86:38–48
Drews G (1965) Untersuohungen zur Regulation der Bacteriochlorophyll-Synthese bei Rhodospirillum rubrum. Archiv Mikrobiologie 51:186–198
Epstein W, Laimins L (1980) Potassium transport in E. coli: diverse systems with common control by osmotic force. Trends Biochem Sci 5:21–23
Fuchs RL, Keister DL (1980) Comparative properties of glutamine synthetase I and II in Rhizobium and Agrobacterium Sp. J Bacteriol 144:648–688
Graham PH, Parker CA (1964) Diagnostic features in the characterisation of root nodule bacteria of legumes. Plant Soil 20:383–396
Herbert D, Philips J, Strange RE (1971) Chemical analysis of microbial cells. Meth Microbiol 5B:210–344
Hua SST, Tsai VY, Lichens GM, Noma AT (1982) Accumulation of amino acids in Rhizobium Spp. strain WR1001 in response to sodium chloride salinity. App Environ Microbiol 44:135–140
Kobayashi H (1982) Second system for potassium transport in Streptococcus faecalis. J Bacteriol 150:506–511
Lim ST, Shanmugam KT (1979) Regulation of hydrogen utilisation in Rhizobium japonicum by cyclic AMP. Biochim Biophys Acta 584:479–492
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with Folin-phenol reagent. J Biol Chem 193:265–272
Measures JC (1975) Role of amino acids in osmoregulation of nonhalophilic bacteria. Nature (Lond) 257:398–400
Prusiner S, Miller RW, Valentine RC (1972) Adenosine 3′5′-cyclic monophosphate control of the enzyme of glutamine metabolism. Proc Natl Acad Sci USA 69:2922–2926
Rains DW (1979) Salt tolerance of plants: strategies of biological systems. In: Hollander A et al. (eds) The biosaline concept. Plenum Press, New York, pp 47–67
Rhoads DB, Epstein W (1977) Energy coupling to net K+ transport in Escherichia coli K-12. J Biol Chem 252:1394–1401
Shapiro BM, Stadtman ER (1970) Glutamine synthetase (E. coli). Meth Enzymol 17A:910–922
Steinborn J, roughley RJ (1974) Sodium chloride as a cause of low numbers of Rhizobium in legume inoculants. J Appl Microbiol 37:93–99
Tempest DW, Meers JL (1970) Influence of environment on content and composition of microbial free amino acid pool. J Gen Microbiol 64:171–185
Yadav NK, Vyas SR (1963) Salt and pH tolerance of Rhizobia. Folia Microbiol 18:242–247
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yap, S.F., Lim, S.T. Response of Rhizobium sp. UMKL 20 to sodium chloride stress. Arch Microbiol 135, 224–228 (1983). https://doi.org/10.1007/BF00414484
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00414484