Abstract
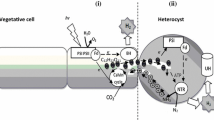
A comparative study has been made on the pigment composition and nitrogenase activity of whole filaments and isolated beterocysts from a mutant strain of Anabaena CA. The whole cell absorption spectra of intact filaments and isolated heterocysts showed close resemblance especially between 550–700 nm region. On a quantitative basis the chlorophyll a content was found almost equal between the vegetative cell and heterocyst but the c-phycocyanin content in the heterocyst was about 1/2 that of the vegetative cell. The purification of the phycobiliprotein on DEAE-cellulose showed the presence of c-phycocyanin (γmax 615 nm) and allophycocyanin (γmax 645 nm, shoulder 620 nm). Isolated heterocysts under H2 showed acetylene reduction rates of 57 nmol C2H4/mg dry wt·min (342 μmol C2H4/mg chl a·h), whereas intact filaments reduced at the rate of 18 nmol C2H4/mg dry wt·min (108 μmol C2H4/mg chl a·h). This rate accounts for 30% recovery of nitrogenase activity in isolated heterocysts compared to whole filaments. The activity was strictly light dependent and was linear under H2 for more than 3 h. Addition of as little as 5% H2 under argon stimulated the C2H2 reductionseveral fold. The acetylene reduction (nitrogenase activity) also showed tolerance to 5% added O2 either under H2 or argon. The results suggest that the heterocyst of Anabaena CA-V is different in some characteristics (viz., higher endogenous C2H2 reduction rate, prolonged activity and higher levels of phycobiliproteins) than those reported in other Anabaena species.
Similar content being viewed by others
References
Alberte RS, Tel-Or E, Packer L, Thornber JP (1980) Functional organization of the photosynthetic apparatus in heterocysts of nitrogen-fixing cyanobacteria. Nature 284:481–483
Bottomley PJ, Van Baalen C (1978) Dark hexose metabolism by photoautotrophically and heterotrophically grown cells of the bluegreen alga (cyanobacterium) Nostoc sp Mac. J Bacteriol 135:888–894
Bradley S, Carr NG (1976) Heterocysts and nitrogenase development in Anabaena cylindrica. J Gen Microbiol 96:175–184
Bryant DA, Glazer AN, Eiserling FA (1976) Characterization and structural properties of the major biliproteins of Anabaena sp. Arch Microbiol 110:61–75
Eisbrenner G, Bothe H (1979) Modes of electron transfer from molecular hydrogen in Anabaena cylindrica. Arch Microbiol 123:37–45
Eisbrenner G, Distler E, Floener L, Bothe H (1978) The occurrence of the hydrogenase in some blue-green algae. Arch Microbiol 118:177–184
Fay P (1969) Cell differentiation and pigment composition in Anabaena cylindrica. Arch Microbiol 67:62–70
Fay P (1970) Photostimulation of nitrogen fixation in Anabaena cylindrica. Biochim Biophys Acta 216:353–356
Fay P (1980) Heterocyst isolation. In: San Pietro A (ed) Methods in enzymology, vol 69C. Academic Press Inc., New York London, pp 801–812
Fay P, Lang NJ (1971) The heterocysts of blue-green algae. 1. Ultrastructural integrity after isolation. Proc R Soc Ser B (Lond) 178:185–192
Fay P, Walsby AE (1966) Metabolic activities of isolated heterocysts of the blue-green alga Anabaena cylindrica. Nature 209:94–95
Fleming H, Haselkorn R (1973) Differentiation in Nostoc muscorum: nitrogenase is synthesized in heterocysts. Proc Natl Acad Sci USA 70:2727–2731
Fogg GE (1949) Growth and heterocyst production in Anabaena cylindrica Lemm. II. In relation to carbon and nitrogen metabolism. Ann Bot (Lond) 13:241–259
Giddings TH, Wolk CP (1981) Factors influencing the stability of nitrogenase activity in isolated cyanobacterial heterocysts. FEMS Microbiol Lett 10:299–302
Gotto JW, Tabita FR, Van Baalen C (1979) Mutants of Anabaena strain CA altered in their ability to grow under nitrogen-fixing conditions. J Bacteriol 140:327–332
Gotto JW, Tabita FR, Van Baalen C (1979) Isolation and characterization of rapidly-growing, marine, nitrogen-fixing strains of blue-green algae. Arch Microbiol 121:155–159
Haselkorn R (1978) Heterocysts. Ann Rev Plant Physiol 29:319–344
Lambert GR, Smith GD (1981) The hydrogen metabolism of cyanobacteria (blue green algae). Biol Rev 56:589–660
Lockau W, Peterson RB, Wolk CP, Burris RH (1978) Modes of reduction of nitrogenase in heterocysts isolated from Anabaena species. Biochim Biophys Acta 502:298–308
Lugtenberg B, Meljers J, Peters R, van der Hoeck P, van Alpen L (1975) Electrophoretic resolution of the major outer membrane protein of Escherichia coli K12 into four bands. FEBS Lett 58:254–258
MacLeod RA (1965) The question of the existence of specific marine bacteria. Bacteriol Rev 29:9–23
Pearce SM, Hildebrandt VA, Lee T (1977) Third system for neutral amino acid transport in a marine pseudomonad. J Bacteriol 130:37–47
Peterson RB, Burris RH (1976) Properties of heterocysts isolated with colloidal silica. Arch Microbiol 108:35–40
Peterson RB, Dolan E, Calvert HE, Ke B (1981) Energy transfer from phycobiliproteins to photosystem I in vegetative cells and heterocysts of Anabaena variabilis. Biochim Biophys Acta 634:237–248
Peterson RB, Wolk CP (1978) High recovery of nitrogenase activity and of 55Fe-labeled nitrogenase in heterocysts isolated from Anabaena variabilis. Proc Natl Acad Sci USA 75:6271–6275
Stacey G, Van Baalen C, Tabita FR (1977) Isolation and characterization of a marine Anabaena sp. capable of rapid growth on molecular nitrogen. Arch Microbiol 114:197–201
Stewart WDP (1980) Some aspects of structure and function in N2-fixing cyanobacteria. Ann Rev Microbiol 34:497–536
Stewart WDP, Haystead A, Pearson HW (1969) Nitrogenase activity in heterocysts of blue-green algae. Nature 224:226–228
Tel-Or E, Stewart WDP (1976) Photosynthetic electron transport, ATP synthesis, and nitrogenase activity in isolated heterocysts of Anabaena cylindrica. Biochim Biophys Acta 423:189–195
Thomas J (1970) Absence of the pigments of photosystem II of photosynthesis in heterocysts of a blue-green alga. Nature 228:181–183
Thomas J, Meeks JC, Wolk CP, Shaffer PW, Austin SM, Chien WS (1977) Formation of glutamine from [13N] ammonia, [13N] dinitrogen, and [14C] glutamate by heterocysts isolated from Anabaena cylindrica. J Bacteriol 129:1545–1555
Van Baalen C (1980) Mutants of blue-green algae. In: San Pietro A (ed) Methods in enzymology, vol 69C. Academic Press Inc., New York London, pp 38–47
Wolk CP (1968) Movement of carbon from vegetative cells to heterocysts in Anabaena cylindrica. J Bacteriol 96:2138–2143
Wolk CP, Simon RD (1969) Pigments and lipids of heterocysts. Planta 86:92–97
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumar, A., Tabita, F.R. & Van Baalen, C. Isolation and characterization of heterocysts from Anabaena sp. strain CA. Arch. Microbiol. 133, 103–109 (1982). https://doi.org/10.1007/BF00413520
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00413520