Abstract
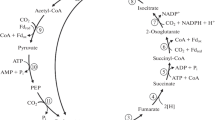
Pathways of succinyl-Coenzyme A (succinyl-CoA) formation in various photosynthetic bacteria were investigated through several approaches, including determination of activity levels of relevant enzymes. Extracts of photosynthetically grown cells of representative Rhodospirillaceae and Chromatium vinosum showed α-ketoglutarate dehydrogenase (KGD) activities sufficient to account for generation of the succinyl-CoA required for biosynthetic metabolism. Except as noted below, the observed ratios of fumarate reductase/succinate dehydrogenase activities were low, consistent with the conclusion that these organisms produce succinyl-CoA oxidatively from α-ketoglutarate (KG), rather than by reductive metabolism of fumarate. On the other hand, the green bacterium Chlorobium limicola appears to produce succinyl-CoA by the reductive pathway; in this organism, KGD activity could not be detected, and a high fumarate reductase/succinate dehydrogenase ratio was observed. Results obtained with Rhodopseudomonas gelatinosa suggest that this otherwise typical member of the Rhodospirillaceae may be able to generate succinyl-CoA via both “arms” of the citric acid cycle, that is, oxidatively from KG, and reductively from fumarate. To further explore the several physiological roles of the conversion: KG→succinyl-CoA in Rhodopseudomonas capsulata, a mutant (strain KGD 11) almost completely blocked in KGD activity was isolated and studied in detail. Under anaerobic photosynthetic conditions, KGD 11 grows readily on succinate as the sole carbon source; in contrast to the wild type parent, however, it cannot grow with l-glutamate as the source of carbon. The R. capsulata parental strain can grow in darkness as an aerobic heterotroph on various carbon/energy sources including pyruvate, D,L-malate, or succinate. Mutant KGD 11, however, is unable to grow aerobically on the substrates noted. These results indicate that the energy for aerobic dark growth of R. capsulata is provided by ”respiratory phosphorylation” fueled by citric acid cycle function, and that this requires a substantial level of KGD activity. The present findings also indicate that citric acid cycle sequences in most of the Rhodospirillaceae prominently used in current research are geared to operate in the oxidative direction, as in nonphotosynthetic aerobic heterotrophs.
Similar content being viewed by others
Abbreviations
- CoA:
-
coenzyme A
- FR:
-
fumarate reductase
- KG:
-
α-ketoglutarate
- KGD:
-
α-ketoglutarate dehydrogenase
- SD:
-
succinate dehydrogenase
References
Allison MJ, Robinson IM, Baetz AL (1979) Synthesis of α-ketoglutarate by reductive carboxylation of succinate in Veillonella, Selenomonas, and Bacteroides species. J Bacteriol 140:980–986
Arrigoni O, Singer TP (1962) Limitations of the phenazine methosulphate assay for succinic and related dehydrogenases. Nature 193:1256–1258
Bose SK, Gest H (1962) Hydrogenase and light-stimulated electron transfer reactions in photosynthetic bacteria. Nature 195:1168–1171
Cox JC, Madigan MT, Favinger JL, Gest H (1980) Redox mechanisms in “oxidant-dependent” hexose fermentation by Rhodopseudomonas capsulata. Arch Biochem Biophys 204:10–17
Dawson RMC, Elliott DC, Elliott WH, Jones KM, (eds) (1969) Data for biochemical research. Oxford University Press, New York Oxford, pp 196–197
Fuchs G, Stupperich E, Thauer RK (1978) Function of fumarate reductase in methanogenic bacteria (Methanobacterium). Arch Microbiol 119:215–218
Fuller RC, Smillie RM, Sisler EC, Kornberg HL (1961) Carbon metabolism in Chromatium. J Biol Chem 236:2140–2149
Gehring U, Arnon DI (1972) Purification and properties of α-ketoglutarate synthase from a photosynthetic bacterium. J Biol Chem 247:6963–6969
Gest H (1980) The evolution of biological energy-transducing systems. FEMS Microbiol Lett 7:73–77
Hillmer P, Gest H (1977) H2 metabolism in the photosynthetic bacterium Rhodopseudomonas capsulata: H2 production by growing cultures. J Bacteriol 129:724–731
Johansson BC, Gest H (1977) Adenylylation/deadenylylation control of the glutamine synthetase of Rhodopseudomonas capsulata. Eur J Biochem 81:365–371
Jones RW, Garland PB (1977) Sites and specificity of the reaction of bipyridylium compounds with anaerobic respiratory enzymes of Escherichia coli. Effects of permeability barriers imposed by the cytoplasmic membrane. Biochem J 164:199–211
Kornfeld S, Benziman M, Milner Y (1977) α-Ketoglutarate dehydrogenase complex of Acetobacter xylinum. Purification and regulatory properties. J Biol Chem 252:2940–2947
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Madigan MT, Gest H (1979) Growth of the photosynthetic bacterium Rhodopseudomonas capsulata chemoautotrophically in darkness with H2 as the energy source. J Bacteriol 137:524–530
Madigan MT, Cox JC, Gest H (1980) Physiology of dark fermentative growth of Rhodopseudomonds capsulata. J Bacteriol 142:908–915
Meynell GG, Meynell E (1970) Theory and practice in experimental bacteriology. Cambridge University Press, Cambridge, pp 257–259
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York, pp 130–134
Niel CB van (1971) Techniques for the enrichment, isolation and inaintenance of the photosynthetic bacteria. In: San Pietro A (ed) Methods in Enzymology, vol 23A, Academic Press, New York London, pp 3–28
Parker MG, Weitzman PDJ (1973) The purification and regulatory properties of α-oxoglutarate dehydrogenase from Acinetobacter lwoffi. Biochem J 135:215–223
Peck HD Jr, Smith OH, Gest H (1957) Comparative biochemistry of the biological reduction of fumaric acid. Biochim Biophys Acta 25:142–147
Pfennig N (1965) Anteicherungskulturen für rote und grüne Schwefelbakterien. Zentbl Bakteriol Parasitenkde Infektionskr-Hyg Abt 1, Suppl 1:179–189
Reed LJ, Mukherjee BB (1969) α-Ketoglutarate dehydrogenase complex from Escherichia coli. In: Lowenstein JM (ed) Methods in Enzymology. vol 13, Academic Press New York London, pp 55–61
Spencer ME, Guest JR (1973) Isolation and properties of fumarate reductase mutants of Escherichia coli. J Bacteriol 114:563–570
Thauer RK, Jungermann K, Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev 41:100–180
Wall JD, Weaver PF, Gest H (1975) Gene transfer agents, bacteriophages, and bacteriocins of Rhodopseudomonas capsulata. Arch Microbiol 105:217–224
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Beatty, J.T., Gest, H. Generation of succinyl-coenzyme A in photosynthetic bacteria. Arch. Microbiol. 129, 335–340 (1981). https://doi.org/10.1007/BF00406457
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00406457