Abstract
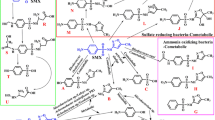
The yield of Hyphomicrobium EG on dimethyl sulphoxide, dimethyl sulphide and methylamine, considering the metabolic pathways of these compounds, suggested that the organism gained energy from the oxidation of the sulphur moiety of the former compounds. Indeed, a comparison of chemostat cultures of Hyphomicrobium EG grown on methylamine in the presence and absence of sulphide or thiosulphate proved this obligate methylotroph to be a chemolithoheterotroph. The apparent Ysulphide and Ythiosulphate were comparable, being 8–10 g dry weight/mol. In batch cultures thiosulphate concentrations up to 10 mM had a stimulatory effect on the growth rate of Hyphomicrobium EG, whereas higher concentrations increased the organisms doubling time.
Enzyme- and respiration data showed that the organism had constitutive enzymes for the breakdown of dimethyl sulphoxide although they were clearly regulated to need. Addition of sulphide or thiosulphate to methylamine-limited chemostat cultures of Hyphomicrobium EG not only resulted in the induction of enzymes necessary for their breakdown, but also caused the enzymes for dimethyl sulphoxide metabolism, especially methyl mercaptan oxidase, to be induced. The formation of H2O2, a product of the latter enzyme, was reflected in the relatively high catalase activities during growth on dimethyl sulphoxide and in the organisms inability to grow on this compound in the presence of a catalase inhibitor.
Similar content being viewed by others
Abbreviations
- DMSO:
-
dimethyl sulphoxide
- DMS:
-
dimethyl sulphide
- MM:
-
methyl mercaptan
- TMAO:
-
trimethylamine N-oxide
- D:
-
dilution rate
- GSH:
-
redticed glutathione
- DCPIP:
-
2,6-dichlorophenolindophenol
- PMS:
-
phenazine methosulphate
- PES:
-
phenazine ethosulphate
- RubPCase:
-
ribulose 1,5-bisphosphate carboxylase
- PEPCase:
-
phosphoenol pyruvate carboxylase
- Wurster's blue (TMPD):
-
N,N,N′,N′-tetramethyl-p-phenylenediamine
References
Anthony C (1982) The biochemistry of methylotrophs. Academic Press. New York London
Attwood MM, Harder W (1977) Isocitrate lyase activity in Hyphomicrobium spp: a critical reappraisal. FEMS Microbiol Lett 1:25–30
Bamforth CW, Large PJ (1977a) Solubilization, partial purification and properties of N-methylglutamate dehydrogenase from Pseudomonas aminovorans. Biochem J 161:357–370
Bamforth CW, Large PJ (1977b) The molecular size of N-methylglutamate dehydrogenase of Pseudomonas aminovorans. Biochem J 167:509–512
Bamforth CW, O'Connor ML (1979) The isolation of pleiotropic mutants of Pseudomonas aminovorans deficient in the ability to grow on methylamine and an examination of their enzymic constitution. J Gen Microbiol 110:143–149
Battat E, Goldberg I, Mateles RI (1974) Growth of Pseudomonas C on C1 compounds: continuous culture. Appl Microbiol 28:906–911
Beudeker RF, Cannon GC, Kuenen JG, Shively JM (1980) Relations between D-ribulose-1,5-bisphosphate carboxylase, carboxysomes and CO2-fixing capacity in the obligate chemolithotroph Thiobacillus neapolitanus grown under different limitations in the chemostat. Arch Microbiol 124:185–189
van Dijken JP, de Bont JAM, Harder W (1981) Dimethyl sulphoxide and dimethyl sulphide as a carbon, sulphur and energy source for growth of Hyphomicrobium S. J Gen Microbiol 127:315–323
Boulton CA, Large PJ (1977) Synthesis of certain assimilatory and dissimilatory enzymes during bacterial adaptation to growth on trimethylamine. J Gen Microbiol 101:151–156
Chen KY, Asce AM, Morris JC (1972) Oxidation of sulfide by O2: catalysis and inhibition. J San Eng Div 98:215–227
Cohen G, Somerson NL (1969) Catalase-aminotriazole method for measuring secretion of hydrogen peroxide by microorganisms. J Bacteriol 98:543–546
Cox RB, Quayle JR (1975) The autotrophic growth of Micrococcus denitrificans on methanol. Biochem J 150:569–571
Dixon GH, Kornberg HL (1959) Assay methods for key enzymes of the glyoxylate cycle. Biochem J 72:3
Eady RR, Large PJ (1968) Purification and properties of an amine dehydrogenase from Pseudomonas AM1 and its role in growth on methylamine. Biochem J 106:245–255
Friedrich CG, Mitrenga G (1981) Oxidation of thiosulfate by Paracoccus denitrificans and other hydrogen bacteria. FEMS Microbiol Lett 10:209–212
Goa J (1953) A microbiuret for protein determination; determination of total protein in cerebrospinal fluid. Scand J Clin Lab Invest 5:218
Gottschal JC, Kuenen JG (1980a) Mixotrophic growth of Thiobacillus A2 on acetate and thiosulphate as growth limiting substrates in the chemostat. Arch Microbiol 126:33–42
Gottschal JC, Kuenen JG (1980b) Selective enrichment of facultatively chemolithotropic thiobacilli and related organisms in the chemostat. FEMS Microbiol Lett 7:241–247
Harder W, Quayle JR (1971) The biosynthesis of serine and glycine in Pseudomonas AM1 with special reference to growth on carbon sources other than C1 compounds. Biochem J 121:753–762
Houle MJ, Long DE, Smette D (1970) A simplex optimized colorimetric method for formaldehyde. Anal Lett 3:401–409
Johnson A, Quayle JR (1964) Microbial growth on C1-compounds. VI Oxidation of methanol, formaldehyde and formate by methanol grown Pseudomonas AM1. Biochem J 93:281–290
Kanagawa T, Kelly DP (1986) Breakdown of dimethyl sulphide by mixed cultures and by Thiobacillus thioparus. FEMS Microbiol Lett 34:13–19
Katayama-Fujimura Y, Kuraishi H (1983) Emendation of Thiobacillus perometabolis London and Rittenberg 1967. Int J Syst Bacteriol 33:650–651
Kelly DP, Kuenen JG (1984) Ecology of the colourless sulphur bacteria. In: Codd GA (ed) Aspects of microbial metabolism and ecology. Academic Press, New York London, pp 211–240
Kelly DP, Wood AP (1982) Autotrophic growth of Thiobacillus A2 on methanol. FEMS Microbiol Lett 15:229–233
Kuwata K, Uebori M, Yamada K, Yamazaki Y (1982) Liquid chromatographic determination of alkylthiols via derivatization with 5,5′-dithiobis (2-nitrobenzoic acid). Anal Chem 54:1082–1087
Large PJ, Peel D, Quale JR (1962) Microbial growth on C1 compounds. 4. Carboxylation of phosphoenolpyruvate in methanol-grown Pseudomonas AM1. Biochem J 85:243–250
Large PJ, Quayle JR (1963) Microbial growth on C1-compounds. 5. Enzyme activities in extracts of Pseudomonas AM1. Biochem J 87:386–396
Lettl A (1984) Soil heterotrophic bacteria in transformations of inorganic sulphur. Folia Microbiol 29:131–137
Levering PR, Dijkhuizen L, Harder W (1984) Metabolic regulation in the facultative methylotroph Arthrobacter P1. Growth on primary amines as carbon and energy sources. Arch Microbiol 139:188–195
London J, Rittenberg SC (1967) Thiobacillus perometabolis nov. sp., a non-autotrophic Thiobacillus. Arch Mikrobiol 59:218–255
Lück H (1963) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press, New York London, pp 885–894
Ochoa S (1948) Biosynthesis of tricarboxylic acids by carbon dioxide fixation. III Enzymatic mechanisms. J Biol Chem 174:133–157
Ochoa S (1955) Malic dehydrogenase from pig heart. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 1. Academic Press, London, pp 735–739
Patel RN, Hoare DS (1971) Physiological studies of methane and methanol oxidizing bacteria: oxidation of C1-compounds by Methylococcus capsulatus. J Bacteriol 107:187–192
Perez RC, Matin A (1980) Growth of Thiobacillus novellus on mixed substrates (mixotrophic growth). J Bacteriol 142:633–638
Quayle JR (1975) Unsolved problems in the microbial metabolism of methane and methanol. In: Terui G (ed) Microbial growth on C1-compounds. The Society of Fermentation Technology, Japan, pp 59–65
Rittenberg SC (1969) The roles of exogenous organic matter in the physiology of chemolithotrophic bacteria. Adv Microb Physiol 3:159–196
Ruby EG, Wirsen CO, Jannasch HW (1981) Chemolithotrophic sulfur-oxidizing bacteria from the Galapagos rift hydrothermal vents. Appl Environ Microbiol 42:317–324
Sahl HG, Trüper HG (1977) Enzymes of CO2 fixation in Chromatiaceae. FEMS Microbiol Lett 2:129–132
Sörbo BH (1957) A colorimetric method for the determination of thiosulphate. Biochim Biophys Acta 23:412–417
Starkey RL (1935) Isolation of some bacteria which oxidize thiosulphate. Biochim Biophys Acta 23:412–417
Starkey RL (1935) Isolation of some bacteria which oxidize thiosulfate. Soil Sci 39:197–219
Stirling DJ, Dalton H (1978) Purification and properties of a NAD(P)+-linked formaldehyde dehydrogenase from Methylococcus capsulatus (Bath). J Gen Microbiol 107:19–29
Suylen GMH, Kuenen JG (1986) Chemostat enrichment and isolation of Hyphomicrobium EG, a dimethyl sulphide oxidizing methylotroph and reevaluation of Thiobacillus MS1. Antonie van Leeuwenhoek J Microbiol Serol 52:281–293
Trudinger PA (1967) Metabolism of thiosulfate and tetrathionate by heterotrophic bacteria from soil. J Bacteriol 93:550–559
Trüper HG, Schlegel HG (1964) Sulphur metabolism in Thiorhodaceae. 1: Quantitative measurements on growing cells of Chromatium okenii. Antonie van Leeuwenhoek J Microbiol Serol 30:225–238
Tuttle JH, Holmes PE, Jannasch HW (1974) Growth rate stimulation of marine pseudomonds by thiosulphate. Arch Microbiol 99:1–14
Tuttle JH (1980) Organic carbon utilization by resting cells of thiosulphate-oxidizing marine heterotrophs. Appl Environ Microbiol 40:516–521
Uebayasi M, Tomizuka N, Kamibayashi A, Tonomura K (1981) Autotrophic growth of a Hyphomicrobium sp. and its hydrogenase activity. Agric Biol Chem 45:1783–1790
Uebayasi M, Kawamura S, Tomizuka N, Kamibayashi A (1985) Comparison of the key enzymes of Hyphomicrobium sp. 53-49 under various growth conditions: aerobic, denitrifying and autotrophic. Agric Biol Chem 49:1799–1807
Vishniac W, Santer M (1957) The Thiobacilli. Bacteriol Rev 21:195–213
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Suylen, G.M.H., Stefess, G.C. & Kuenen, J.G. Chemolithotrophic potential of a Hyphomicrobium species, capable of growth on methylated sulphur compounds. Arch. Microbiol. 146, 192–198 (1986). https://doi.org/10.1007/BF00402350
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00402350