Abstract
It is now generally recognized that cell growth conditions in nature are often suboptimal compared to controlled conditions provided in the laboratory. Natural stresses like starvation and acidity are generated by cell growth itself. Other stresses like temperature or osmotic shock, or oxygen, are imposed by the environment. It is now clear that defense mechanisms to withstand different stresses must be present in all organisms. The exploration of stress responses in lactic acid bacteria has just begun. Several stress response genes have been revealed through homologies with known genes in other organisms. While stress response genes appear to be highly conserved, however, their regulation may not be. Thus, search of the regulation of stress response in lactic acid bacteria may reveal new regulatory circuits. The first part of this report addresses the available information on stress response in Lactococcus lactis.
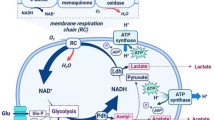
Acid stress response may be particularly important in lactic acid bacteria, whose growth and transition to stationary phase is accompanied by the production of lactic acid, which results in acidification of the media, arrest of cell multiplication, and possible cell death. The second part of this report will focus on progress made in acid stress response, particularly in L. lactis and on factors which may affect its regulation. Acid tolerance is presently under study in L. lactis. Our results with strain MG1363 show that it survives a lethal challenge at pH 4.0 if adapted briefly (5 to 15 minutes) at a pH between 4.5 and 6.5. Adaptation requires protein synthesis, indicating that acid conditions induce expression of newly synthesized genes. These results show that L. lactis possesses an inducible response to acid stress in exponential phase.
To identify possible regulatory genes involved in acid stress response, we determined low pH conditions in which MG1363 is unable to grow, and selected at 37°C for transposition insertional mutants which were able to survive. About thirty mutants resistant to low pH conditions were characterized. The interrupted genes were identified by sequence homology with known genes. One insertion interrupts ahrC, the putative regulator of arginine metabolism; possibly, increased arginine catabolism in the mutant produces metabolites which increase the pH. Several other mutations putatively map at some step in the pathway of (p)ppGpp synthesis. Our results suggest that the stringent response pathway, which is involved in starvation and stationary phase survival, may also be implicated in acid pH tolerance.
Similar content being viewed by others
References
Abdelal AT (1979) Arginine catabolism by microorganisms. Ann. Rev. Microbiol. 33: 139–168.
Araya T, Ishibashi N, Shimamura S, Tanaka K & H Takahashi (1993) Genetic and molecular analysis of the rpoD gene from Lactococcus lactis. Biosci. Biotech. Biochem. 57: 88–92
Arnau J, Sorensen KI, Appel KF, Vogensen FK & K Hammer (1996) Analysis of heat shock gene expression in Lactococcus lactis MG1363. Microbiology, 142: 1685–1691
van Asseldonk M, Simons A, Vissier H, de Vos WM & G Simons (1993) Cloning, nucleotide sequence, and regulatory analysis of the Lactococcus dnaJ gene. J. Bacteriol. 175: 1637–1644
Baumberg S & U Klingel (1993) Biosynthesis of arginine, proline, and related compounds. In: Bacillus subtilis and Other Gram-Positive Bacteria. pp. 299–306 In: AL Sonenshein, R Losick, JA Hoch (Eds.), ASM press
Boutibonnes P, Gillot B, Auffray Y & B Thammavongs (1991) Heat shock induces thermotolerance and inhibition of lysis in a lysogenic strain of Lactococcus lactis. Int. J. Food Microbiol. 14: 1–9
Burland V, G PlunkettIII, DL Daniels & FR Blattner (1993) DNA sequence and analysis of 136 kilobases of the Escherichia coli genome: organizational symmetry around the origin of replication. Genomics 16: 551–561
Cashel M & K Rudd (1987) The stringent response, pp. 1410–1438 In: FC Neidhardt, JL Ingraham, KB Low, B Maganasik, M Schaechter and HE Umbarger (Ed.), Escherichia coli and Salmonella typhimurium: Cellular and Molecular Biology, Vol. 2. American Society for Microbiology, Washington, D.C.
Casiano-Colon A & RE Marquis (1988) Role of the arginine deiminase system in protecting oral bacteria and an enzymatic basis for acid tolerance. Appl. Environ. Microbiol. 54: 1318–1324
Duwat P, SD Ehrlich & A Gruss (1995) The recA gene of Lactococcus lactis: Characterization and involvement in oxidative and thermal stress. Mol. Microbiol. 17: 1121–1131
Duwat P, S Sourice, SD Ehrlich & A Gruss (1995) The recA gene involvement in oxidative and thermal stress in Lactococcus lactis. In “Genetics of Streptococci, Enterococci and Lactococci’. Ferretti JJ, Gilmore MS, Klaenhammer TR & Brown F (Eds.), Dev. Biol. Stand. Basel Karger, publ. Vol 85. pp 455–467
Eaton T., C. Shearman & M. Gasson. 1993. Cloning and sequence analysis of the dnaK region of L. lactis subsp. lactis. J. Gen. Microbiol. 139: 3253–3264
Fang FC, Libby SJ, Buchmeir NA, Loewen PC, Switala J, Harwood J & D.G. Guiney. (1992) The alternative σ factor KatF (RpoS) regulates Salmonella virulence. Proc. Natl. Acad. Sci. USA. 89: 11978–11982
Foster JW & HK Hall (1991) Inducible pH homeostasis and the acid tolerance response of Salmonella typhimurium. J. Bacteriol. 173: 5129–5135
Foster JW (1993) The acid tolerance response of Salmonella typhimurium involves transient synthesis of key acid shock proteins. J. Bacteriol. 175: 1981–1987
Gansel X, Hartke A, Boutibonnes P & Y Auffray (1993) Nucleotide sequence of the Lactococcus lactisNCDO 763 (ML3) rpoD gene. Biochem. et Biophysica Acta 1216: 115–118
Gasson M (1983) Plasmid complements of Streptococcus lactis NCDO712 and other lactic Streptococci after protoplast-induced curing. J. Bacteriol. 154: 1–9
Gentry DR, Hermandez VJ, Nguyen LH, Jensen DB & M Cashel (1993) Synthesis of the stationary-phase sigma factor ss is positively regulated by ppGpp. J. Bacteriol. 175: 7982–7989
Georgopoulos C & WJ Welch (1993) Role of the major heat shock proteins as molecular chaperones. Ann. Rev. Cell. Biol. 9: 601–634
Hall HK, Karem KL & JW Foster (1995) Molecular responses of microbes to environmental pH stress. Adv. Microbial Physiol. 37: 229–272
Hartke A, S Bouche, X Gansel, P Boutibonnes & Y Auffray (1994) Starvation-induced stress resistance in Lactococcus lactis subsp. lactis IL 1403. Appl. Environ. Microbiol. 60: 3474–3478
Hartke A, Giard JC, Benachour A & Y Auffray (1995) The acid-stress response of Lactococcus lactis subsp. lactis IL 1403: Changes in protein synthesis and cross-protection. 7eme Colloque du Club des Bactéries Lactiques Paris 13–15 Septembre 1995. Aff. S5
Hartke A, Bouché S, Boutibonnes P & Y Auffray (1996) UV-inducible proteins and UV-induced Cross-protection against acid, ethanol, H2O2 or heat treatments in Lactococcus lactis subsp. lactis (submitted).
Hecker M, Schumann W & U Völker (1996) Heat-shock and general stress response in Bacillus subtilis. Mol. Microbiol. 19: 417–428
Hengge-Aronis R (1993) The role of rpoS in early stationary-phase gene regulation in Escherichia coli K12. pp. 171–200 In: “Starvation in Bacteria” S Kellenberg (Ed.), Plenum Press
Herman C, Ogura T, Tomoyasu T, Hirage S, Akiyama Y, Ito K, Thomas R, D'Ari R & P Bouloc (1993) Cell growth and λ phage development controlled by the same essential Escherichia coli gene ftsH/hflB. Proc. Natl. Acad. Sci. USA 90: 10861–10865
Herman C, D Thévenet, R D'Ari & P Bouloc (1995) Degradation of σ32, the heat shock regulator in Escherichia coli, is governed by HflB. Proc. Natl. Sci. USA. 92: 3516–3520
Huang DC, Novel M, Huang XF & G Novel (1992) Nonidentity between plasmid and chromosomal copies of ISS1-like sequences in Lactococcus lactis subsp. lactis CNRZ270 and their possible role in chromosomal integration of plasmid genes. Gene 118, 39–46
Israelson H, Madsen S, Vrang A, Hansen E & E Johansen (1995) Cloning and partial characterization of regulated promoters from Lactococcus lactis Tn917-lacZ integrants with the new promoter probe vector pAK80. Appl. and Environ. Microbiol. 61: 2540–2547
Jensen PR & K Hammer. (1993) Minimal requirements for exponential growth of Lactococcus lactis; Appl. Environ. Microbiol. 59: 4363–4366
Kashket ER (1987) Bioenergetics of lactic acid bacteria: cytoplasmic pH and osmotolerance. FEMS Microbiol. Rev. 46: 233–244
Kellenberg S. Starvation in Bacteria. Plenum Press. The reader is referred particularly to chapters 6 through 10
Kihara A, Akiyama Y & K Ito (1995) FtsH is required for proteolytic elimination of uncomplexed forma of Sec Y, an essential protein translocase subunit. Proc. Natl. Acad. Sci. USA 92: 4532–4536
Kim SG & CA Batt (1993) Cloning and sequencing of the Lactococcus lactis subsp. lactis groESL operon. Gene 127: 121–126
Kolter R (1993) The stationary phase of the bacterial life cycle. Annu. Rev. Biochem. 47: 855–874
Kunji ERS, Ubbink T, Matin A, Poolman B & WN Konings (1993) Physiological responses to Lactococcus lactis ML3 to altering conditions of growth and starvation. Arch. Microbiol. 159: 372–379
Lange R, Fischer D & R Hengge-Aronis (1995) Identification of transcriptional start sites and the role of ppGpp in the expression of rpoS, the structural gene for the ss subunit of RNA polymerase in Escherichia coli. J. Bacteriol. 177: 4676–4680
Law J, Buist G, Haandrikman A, Kok J, Venema G & K Leenhouts (1995) A system to generate chromosomal mutations in Lactococcus lactis which allows fast analysis of targeted genes. J. Bacteriol. 177: 7011–7018
LeBourgeois P, Lautier M, Mata M & P Ritzenthaler (1992) New tools for the physical mapping of Lactococcus lactis strains. Gene 11: 109–114
Lee IS, Slonczewski JL & JW Foster (1994) A low- pH-inducible, stationary-phase acid tolerance response in Salmonella typhimurium. J. Bacteriol. 176: 1422–1426
Lee IS, J Lin, HK Hall, B Bearson & JW Foster (1995) The stationary-phase sigma factor ss (RpoS) is required for a sustained acid tolerance response in virulent Salmonella typhimurium. Mol. Microbiol. 17: 155–167
Loewen PC & R Hengge-Aronis (1994) The role of sima factor σ8 (KatF) in bacterial global regulation. Ann. Rev. Microbiol. 48: 53–80
Maguin E, P Duwat, T Hege, D Ehrlich & A Gruss (1992) New thermosensitive plasmid for Gram-positive bacteria. J. Bacteriol. 174: 5633–5638
Maguin E, H Prévost, SD Ehrlich & A Gruss (1996) Efficient insertional mutagenesis in Lactococci and other Gram-positive bacteria. J. Bacteriol. 178: 931–935
Mãntsälä P & H Zalkin (1992) Cloning and sequence of Bacillus subtilis purA and guaA, involved in the conversion of IMP to AMP and GMP. J. Bacteriol 174: 1883–1890
Marquis RE, GR Bender, DR Murray & A Wong (1987) Arginine deiminase system and bacterial adaptation to acid environments. Appl. Environ. Microbiol. 53: 198–200
Mechold V, Steiner K, Vettermann S & H Malke (1993) Genetic organization of the streptokinase region of the Streptococcus equisimilis H46A chromosome. Mol. Gen. Genet. 241: 129–140
Nilsson D & AA Lauridsen (1992) Isolation of purine auxotrophic mutants of Lactococcus lactis and characterization of the gene hptencoding hypoxanthine guanine phosphoribosyltransferase. Mol. Gen. Genet. 235: 359–364
Nilsson D, AA Lauridsen, T Tomoyasu & T Ogura (1994) A Lactococcus lactis gene encodes a membrane protein with putative ATPase activity that is homologous to the essential Escherichia coli ftsH gene product. Microbiol. 140: 2601–2610
Nystrom T (1993) Global systems approach to the physiology of the starved cell. pp. 129–150. In: “Starvation in Bacteria” S Kellenberg (Ed.), Plenum Press
Parsell DA & S Lindquist (1993) The function of heat- shock proteins in stress tolerance: degradation and reactivation of damaged proteins. Ann. Rev. Genet. 27: 437–496
Piard JC & M Desmazeaud (1991) Inhibiting factors produced by lactic acid bacteria. 1-Oxygen metabolites and catabolism end-products. Lait. 71: 525–541
Rowbury RJ, M Goodson & AD Wallace (1992) The PhoE porin and transmission of the chemical stimulus for induction of acid resistance (acid habituation) in Escherichia coli. J. Appl. Bacteriol. 72: 233–243
Sanders JW, Leenhouts K, Haandrikman AJ, Venema G & J Kok (1995) Stress response in Lactococcus lactis: Cloning, expression analysis, and mutation of the superoxide dismutase gene. J. Bacteriol. 177: 5254–5260
Slonczewski JL (1992) pH-regulated genes in enteric bacteria. ASM News. 58: 140–144
Small P, D Blankenhorn, D Welty, E Zinser & J Slonczewski (1994) Acid and base resistance in Escherichia coli and Shigella flexneri: Role of rpoS and growth pH. J. Bacteriol. 176: 1729–1737
Spira B, Silberstein N & E Yagil (1995) Guanosine 3′, 5′-Bispyrophosphate (ppGpp) synthesis in cells of Escherichia coli starved for Pi. J. Bacteriol. 177: 4053–4058
Ullerup A, Saxild HH & D Nilsson (1995) Regulation of Lactococcus lactis ftsH expression. 7eme Colloque du Club des Bactéries Lactiques Paris 13–15 Septembre 1995. Aff. G8
Visick JE & S Clarke (1995) Repair, refold, recycle: how bacteria can deal with spontaneous and environmental damage to proteins. Mol. Microbiol. 16: 835–845
Yuan G & SL Wong (1995) Isolation and characterization of Bacillus subtilis groE regulatory mutants: Evidence for orf39 in the dnaK operon as a repressor gene in regulating the expression of both groE and dnaK. J. Bacteriol. 177: 6462–6468
Zuber U & W Schuman (1994) CIRCE, a novel heat shock element involved in regulation of heat shock operon dnaK of Bacillus subtilis. 176: 1359–1363
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rallu, F., Gruss, A. & Maguin, E. Lactococcus lactis and stress. Antonie van Leeuwenhoek 70, 243–251 (1996). https://doi.org/10.1007/BF00395935
Issue Date:
DOI: https://doi.org/10.1007/BF00395935