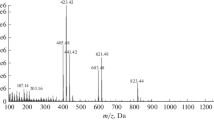
Summary
In the supernatant of melanoma cell culture SK mel 25 three indolic compounds-5-hydroxy-6-methoxyindole, 5-hydroxy-6-methoxyindolyl-2-carboxylic, and 6-hydroxy-5-methoxyindolyl-2-carboxylic acids-have been identified. The supernatants were extracted with ethyl acetate, derivatized, and analyzed by gas chromatography-mass spectrometry. The significance of this finding is discussed.
Similar content being viewed by others
Abbreviations
- 5,6DHI:
-
5,6-dihydroxyindole
- 5H6MI:
-
5-hydroxy-6-methoxyindole
- 6H5MI:
-
6-hydroxy-5-methoxyindole
- 5,6DHI2C:
-
5,6-dihydroxyindolyl-2-carboxylic acid
- 5H6MI2C:
-
5-hydroxy-6-methoxyindolyl-2-carboxylic acid
- 6H5MI2C:
-
6-hydroxy-5-methoxyindolyl-2-carboxylic acid
- PFPA:
-
pentafluoropropionic anhydride
- HFIP:
-
1,1,1,3,3,3-hexafluoroisopropanol
- DOPA:
-
L-3,4-dihydroxyphenylalanine
- COMT:
-
catechol-O-methyltransferase (EC 2.1.1.6)
- GC-MS:
-
gas chromatography-mass spectrometry
References
Axelrod J, Lerner AB (1963) O-methylation in the conversion of tyrosine to melanin. Biochim Biophys Acta 71:650–655
Duchoň J, Matouš B, Pechan Z (1966) On the chemical nature of urinary melanogens. In: Della Porta G, Mühlbock O (eds) Structure and control of the melanocyte. Springer, Berlin Heidelberg New York (pp 175–184)
Duchoň J, Matouš B (1967) Identification of two new metabolites in melanoma urine: 5-hydroxy-6-methoxyindole-2-carboxylic and 5-methoxy-6-hydroxyindole-2-carboxylic acids. Clin Chim Acta 16:397–402
Håkanson R, Möller H, Stomby NG (1965) DOPA decarboxylase activity in a transplantable hamster melanoma. Experimentia 21:265–266
Pavel S, Muskiet FAJ, Nagel GT, Schwippelová Z, Duchoň J (1981a) Identification of two Thormählen-positive compounds from melanotic urine by gas chromatography-mass spectrometry. J Chromatogr (Biomed Appl) 222:329–336
Pavel S, Muskiet FAJ, Buděšínská A, Duchoň J (1981b) A qualitative gas chromatographic analysis of substituted 5,6-dihydroxyindoles from the urine of patient with melanoma. Tumori 67:325–332
Pavel S, Muskiet FAJ (1983) Q Synthesis of deuterium labelled eumelanin (precursor) metabolites. J Lab Comp Radiopharmacol 20:101–110
Pavel S, Muskiet FAJ (1983) Eumelanin (precursor) metabolites as markers for pigmented malignant melanoma. A preliminary report. Canc Det Prev [Suppl] (in press)
Pawelek JM, Lerner AB (1978) 5,6-dihydroxyindole is a melanin precursor showing potent cytotoxicity. Nature 276:627–628
Prota G (1980) Recent advances in the chemistry of melanogenesis in mammals. J Invest Dermatol 75:122–127
Wick MM (1980) An experimental approach to the chemotherapy of melanoma. J Invest Dermatol 74:63–65
Author information
Authors and Affiliations
Additional information
This work was supported by grants from Koningin Wilhelmina Fonds (Netherlands Cancer Foundation) Nos.82-10 and 82-6 GUKC
Rights and permissions
About this article
Cite this article
Pavel, S., Muskiet, F.A.J., de Ley, L. et al. Identification of three indolic compounds in a pigmented-melanoma cell-culture supernatant by gas chromatography-mass spectrometry. J Cancer Res Clin Oncol 105, 275–279 (1983). https://doi.org/10.1007/BF00395758
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00395758