Abstract
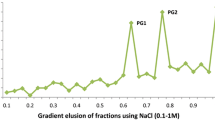
A procedure was developed for the differential extraction of polygalacturonases (PG) I and II from tomatoes (Lycopersicon esculentum Mill.). Extraction of pericarp tissue from ripe fruit at conventional conditions of 1.0 M NaCl and pH 6.0 yielded nearly equal amounts of the two enzymes. However, most of the PG activity could be extracted also with water at pH 1.6, and the water extract contained only PG II. Subsequent extraction of the pellet with 1.0 M NaCl at pH 6.0 and 10.0 yielded some PG I and high levels of PG converter, the protein in tomatoes that reacts with PG II to form PG I. Application of this procedure to tomatoes at different stages of ripening showed that PG II appeared as ripening began and then increased during ripening. Much lower levels of PG I than of PG II were extracted at all stages of ripeness. The PG converter was present in unripe fruit and increased during ripening. The results demonstrate that PG I is formed when PG II and PG converter are solubilized simultaneously and that PG II is the only endogenous PG in tomatoes.
Similar content being viewed by others
Abbreviations
- PG:
-
polygalacturonase
References
Ali, Z.M., Brady, C.J. (1982) Purification and characterization of the polygalacturonases of tomato fruits. Aust. J. Plant Physiol. 9, 155–169
Brady, C.J., McGlasson, W.B., Pearson, J.A., Meldrum, S.K., Kopeliovitch, E. (1985) Interaction between the amount and molecular forms of polygalacturonase, calcium, and firmness in tomato fruit. J. Am. Soc. Hort. Sci. 110, 254–258
Brady, C.J., Meldrum, S.K., McGlasson, W.B., Ali, M. (1983) Differential accumulation of the molecular forms of polygalacturonase in tomato mutants. J. Food Biochem. 7, 7–14
Buescher, R.W., Sistrunk, W.A., Tigchelaar, E.C., Ng, T.J. (1976) Softening, pectolytic activity and storage-life of rin and nor tomato hybrids, HortScience 11, 603–605
Crookes, P.R., Grierson, D. (1983) Ultrastructure of tomato fruit ripening and the role of polygalacturonase isoenzymes in cell wall degradation. Plant Physiol. 72, 1008–1093
Hobson, G.E. (1964) Polygalacturonase in normal and abnormal tomato fruit. Biochem. J. 92, 324–332
Moshrefi, M., Luh, B.S. (1983) Carbohydrate composition and electrophoretic properties of tomato polygalacturonase isoenzymes. Eur. J. Biochem. 135, 511–514
Nelson, N. (1944) A photometric adaptation of the Somogyi method for the determination of glucose. J. Biol. Chem. 153, 375–380
Pressey, R. (1984a) Purification and characterization of tomato polygalacturonase converter. Eur. J. Biochem. 144, 217–221
Pressey, R. (1984b) A rapid method for separating tomato polygalacturonases by HPLO. HortScience 19, 572–573
Pressey, R. (1986a) Extraction and assay of tomato polygalacturonases. Hort Science 21, 490–492
Pressey, R. (1986b) Changes in polygalacturonase isoenzymes and converter in tomatoes during ripening. HortScience 21, 1183–1185
Pressey, R., Avants, J.K. (1973) Two forms of polygalacturonase in tomatoes. Biochim. Biophys. Acta 309, 363–369
Pressey, R., Avants, J.K. (1982) Solubilization of cell walls by tomato polygalacturonases: Effects of pectinesterases. J. Food Biochem. 6, 57–74
Tucker, G.A., Robertson, N.G., Grierson, D. (1980) Changes in polygalacturonase isoenzymes during the ripening of normal and mutant tomato fruit. Eur. J. Biochem. 112, 119–124
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pressey, R. Reevaluation of the changes in polygalacturonases in tomatoes during ripening. Planta 174, 39–43 (1988). https://doi.org/10.1007/BF00394871
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00394871