Abstract
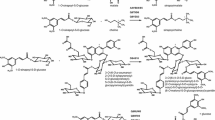
A tigloyl-CoA: 13-hydroxylupanine O-tigloyl-transferase could be demonstrated in crude enzyme preparations from Lupinus albus seedlings. The enzyme activity increases concomitantly with for formation of 13-tigloyloxylupanine in developing lupin seedlings. The transferase catalyzes specifically the transfer of an acyl group to 13-hydroxylupanine. The apparent Km-values are 140 μM for tigloyl-CoA and 18 μM for 13-hydroxylupanine. Other hydroxylated compounds, e.g., lupinine, 4-hydroxylupanine, and cholesterol are not acylated. The transferase shows optimal activity at pH 7–8 and at 30°C. It is activated by dithioerythritol and inhibited by thiol reagents. Tigloyl-CoA can be replaced as acyl donor by benzoyl-CoA and to a lesser extent by valeroyl-CoA, 3-methylbutyryl-CoA, butyryl CoA and propionyl-CoA but not acetyl-CoA. Preliminary evidence indicates that the transfer of the tigloyl and benzoyl moieties is catalyzed by different enzymes. The transferase activity could only be demonstrated in plants which produce quinolizidine alkaloids.
Similar content being viewed by others
Abbreviations
- DIECA:
-
diethyldithiocarbamate
- DTE:
-
dithioerythritol
- GLC:
-
gas-liquid chromatography
References
Chase, J.F.A. (1969) Carnitine acetyltransferase from pigeon breast muscle. Meth. Enzymol. 13, 387–393
Faugeras, G., Paris, R. (1971) Nouvelles recherches phytochimiques sur les Papilionacées-Génistées d'Europe. Boissiera 19, 201–218
Goodman, D.S. (1969) Hydrolysis and formation of cholesterol esters in rat liver. Meth. Enzymol. 15, 522–537
Gross, G.G. (1981) Phenolic acids. In: Biochemistry of plants, vol. 7, pp. 301–316, Stumpf, P.K., Conn, E.E., eds. Academic Press, New York London
Hahlbrock, K. (1981) Flavonoids. In: The biochemistry of plants Vol. 7, pp. 425–456, Stumpf, P.K., Conn, E.E., eds. Academic Press, New York London
Murakoshi, I., Ogawa, M., Toriizuka, K., Haginiwa, J., Ohmiya, S., Otomasu, H. (1977) The enzymic conversion of (-)-lupinine to-(-) trans-(4′-hydroxycinnamoyl)lupinine by extracts of Lupinus seedings. Chem. Pharm. Bull. 25, 527–528
Prince, A.K. (1971) Choline acetyltransferase. Meth. Enzymol. 17B, 788–798
Wink, M., Hartmann, T. (1981) Sites of enzymatic synthesis of quinolizidine alkaloids and their accumulation in Lupinus polyphyllus. Z. Pflanzenphysiol. 102, 337–344
Wink, M., Witte, L., Schiebel, H.M., Hartmann, T. (1980a) Alkaloid pattern of cell suspension cultures and differentiated plants of Lupinus polyphyllus. Planta Med. 38, 238–245
Wink, M., Hartmann, T., Witte, L. (1980b) Biotransformation of cadaverine and potential intermediates of lupanine biosynthesis by plant cell suspension cultures. Planta Med 40, 31–39
Wink, M., Hartmann, R., Witte, L., Schiebel, H.M. (1981a) The alkaloid pattern of cell suspension cultures and differentiated plants of Baptisia australis and their biogenic implications. J. Nat. Prod. 44, 14–20
Wink, M., Witte, L., Hartmann, T., (1981b) Quinolizidine alkaloids of cell suspension cultures and plants of Sarothamnus scoporius and of its root parasite Orobanche rapum-genistae. Planta Med. 43, 342–352
Wink, M., Schiebel, H.M., Witte, L., Hartmann, T. (1982) Quinolizidine alkaloids from cell suspension cultures and plants. Ester alkaloids of Lupinus polyphyllus. Planta Med. 44, 15–20
Zenk, M. (1979) Recent work on cinnamoyl CoA derivatives. Recent Adv. Phytochem. 12, 139–176
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wink, M., Hartmann, T. Enzymatic synthesis of quinolizidine alkaloid esters: a tigloyl-CoA: 13-hydroxylupanine O-tigloyltransferase from Lupinus albus L.. Planta 156, 560–565 (1982). https://doi.org/10.1007/BF00392781
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392781