Abstract
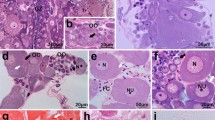
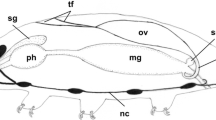
Female kuruma prawns (Penaeus japonicus Bate) with undeveloped, early developing, developing, nearly ripe and ripe ovaries, were collected from Ise Bay, Japan, in 1984. Oocyte development of the kuruma prawn was classified into ten stages according to morphological characters, namely: (1) synapsis stage, (2) chromatin nucleolus stage, (3) early perinucleolus stage, (4) late perinucleolus stage, (5) oil globule Stage I, (6) oil globule Stage II, (7) yolkless stage, (8) yolk granule stage, (9) prematuration stage, and (10) maturation stage. The synapsis stage is a multiplication stage. The chromatin nucleolus stage, early and late perinucleolus stages are previtellogenesis and primary growth stages. Oil globule Stage I is an initial stage of primary vitellogenesis and secondary growth. Follicle cells on the oil globule Stage I oocytes expand rapidly and reach maximum size during oogenesis. Yolk granule stage oocytes are in the initial stages of secondary vitellogenesis. Strongly acidophilic yolk granules accumulate within basophilic vesicles of the cytoplasm. The yolk granules are first concentrated in the inner part of the cytoplasm, then gradually spread to the periphery. Cortical crypts, which are separated from the oocyte cytoplasm by the cytoplasmic membrane, are situated outside of oocyte cytoplasm. Germinal vesicle breakdown (GVBD) is initiated in the late phase of prematuration and continues until the late phase of maturation immediately prior to spawning. At the beginning of the maturation stage, the oocytes are ovulated, after which the nuclei further shrink and migrate out-wards. After ovulation, meiotic division of the ovarian oocyte progressed up to the metaphase of primary maturation division. Finally, the meiotic metaphase is visible just beneath the cytoplasmic membrane in the mature oocyte. Though ovulation is synchronous within the same ovary, GVBD is not completely synchronous. Ovulated mature oocytes have many club-shaped cortical crypts in the peripheral part of the cytoplasm and contain extensive accumulations of yolk granules dispersed throughout the cytoplasm. The apical end of the club-shaped cortical crypts and cytoplasmic membrane are coated by the vitellin envelope in the mature oocyte.
Similar content being viewed by others
Literature cited
Anderson, S. L., Chang, E. S., Clark, W. H. (1984). Timing of postvitellogenic ovarian changes in the ridgeback prawn, Sicyonia ingentis (Penaeidae) determined by ovarian biopsy. Aquaculture, Amsterdam 42: 257–271
Beams, H. W., Kessel, R. G. (1963). Electron microscope studies on developing crayfish oocytes with special reference to the origin of yolk. J. Cell Biol. 18: 621–649
Clark, W. H. Jr., Lynn, J. W., Yudin, A. I., Persyn, H. O. (1980). Morphology of the cortical reaction in the eggs of Penaeus aztecus. Biol. Bull. 158: 175–186
Clark, W. H. Jr., Yudin, A. I., Griffin, F. J., Shigekawa, K. (1984). The control of gamete activation and fertilization in the marine panaeidae, Sicyonic ingentis. In: Engels, W., Clark, W. H., Fischer, A., Olive, P. J. W., Went, D. F. (eds.). Advance in invertebrate reproduction 3. Elsevier Science Publishers B. V., Amsterdam, p. 459–472
Cobb, S. P., Fitch, C. R., Camp, D. K. (1973). The rock shrimp Sicyonia brevirostris, Stimpson, 1871 (Decapoda, Penaeidae). Mem. Hourglass Cruises 3: 1–38
Cummings, W. C. (1961). Maturation and spawning of the pink shrimp, Penaeus duorarum, Burkenroad, Trans. Am. Fish. Soc. 90: 462–468
Duronslet, M. J., Yudin, A. I., Wheeler, R. S., Clark, W. H. (1975). Light and fine structural studies of natural and artificially induced egg grwoth of penaeid shrimp. Proc. World Maricult. Soc. 6: 105–122
Genthe, H. C. (1969). The reproductive biology of Sergestes similis (Decapoda, Natantia). Mar. Biol. 2: 203–217
Hudinaga, M. (1942). Reproduction, development, and rearing of Penaeus japonicus, Bate. Jpn. J. Zool. 10: 305–393
Kennedy, F. S., Crane, J. J., Schlieder, R. A., Barber, D. G. (1977). Studies of the rock shrimp, Sicyonia brevirostris, a new fishery resource on Florida's Atlantic shelf. Fla. Mar. Res. Publ. 27: 69
Kessel, R. G. (1968). Mechanisms of protein yolk synthesis and deposition in crustacean oocytes. Z. Zellforsch. 89: 17–38
King, J. E. (1948). A study of the reproductive organs of the common marine shrimp, Penaeus setiferus, Linnaeus. Biol. Bull. 94: 244–262
O'Connor, C. (1979). Reproductive periodicity of a Penaeus esculentus population near Low Islet, Queensland, Australia. Aquaculture, Amsterdam 16: 153–162
Weitzman, M. C. (1966). Oogenesis in the tropical land crab, Gecarcinus lateralis, Freminville. Z. Zellforsch. 75: 109–119
Yano, I. (1984). Rematuration of spent kuruma prawn, Penaeus japonicus. Aquaculture, Amsterdam 42: 179–183
Yano, I. (1985). Induced ovarian maturation and spawning in greasyback shrimp, Metapenaeus ensis by progesteron. Aquaculture, Amsterdam 47: 223–229
Yano, I. (1987a). Maturation of kuruma prawn Penaeus japonicus cultured in earthen ponds. NOAA Tech. Rep. NMFS 47: 3–7
Yano, I., Chinzei, Y. (1987b). Ovary is the site of vitellogenin synthesis in kuruma prawn, Penaeus japonicus. Comp. Biochem. Physiol. 86: 213–218
Author information
Authors and Affiliations
Additional information
Communicated by M. Anraku, Tokyo
Rights and permissions
About this article
Cite this article
Yano, I. Oocyte development in the kuruma prawn Penaeus japonicus . Mar. Biol. 99, 547–553 (1988). https://doi.org/10.1007/BF00392562
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392562