Abstract
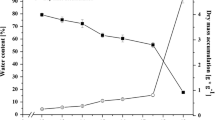
The major storage proteins isolated from wild-type seeds of Arabidopsis thaliana (L.) Heynh., strain “Columbia”, were studied by sucrose gradient centrifugation and sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Both the hypocotyl and cotyledons of mature embryos contained abundant 12 S (cruciferin) and 2 S (arabin) proteins that appeared similar in size and subunit composition to the cruciferin (12 S) and napin (1.7 S) seed-storage proteins of Brassica napus. The 12 S protein from Arabidopsis was resolved by SDS-PAGE into two groups of subunits with approximate relative molecular weights of 22–23 kDa (kilodalton) and 30–34 kDa. These polypeptides accumulated late in embryo development, disappeared early in germination, and were not detected in other vegetative or reproductive tissues. Accumulation of the 12 S proteins in aborted seeds from nine embryo-lethal mutants with different patterns of abnormal development was studied to determine the extent of cellular differentiation in arrested embryos from each mutant line. Abundant 12 S proteins were found in arrested embryos from two mutants with late lethal phases, but not in seven other mutants with lethal phases ranging from the globular to the cotyledon stages of embryo development. These results indicate that the accumulation of seed-storage proteins in wild-type embryos of Arabidopsis is closely tied to morphogenetic changes that occur during embryo development. Embryo-lethal mutants may therefore be useful in future studies on the developmental regulation of storage-protein synthesis.
Similar content being viewed by others
Abbreviations
- kDa:
-
kilodalton
- Mr :
-
relative molecular weight
- PAGE:
-
polyacrylamide gel electrophoresis
- SDS:
-
sodium dodecyl sulfate
References
Baus, A.D., Franzmann, L., Meinke, D.W. (1986) Growth in vitro of arrested embryos from lethal mutants of Arabidopsis thaliana. Theor. Appl. Genet. (in press)
Brown, J.W.S., Ma, Y., Bliss, F.A., Hall, T.C. (1981) Genetic variation in the subunits of globulin-1 storage protein of French bean. Theor. Appl. Genet. 59, 83–88
Burr, F.A., Burr, B. (1982) Three mutations in Zea mays affecting zein accumulation: a comparison of zein polypeptides, in vitro synthesis and processing, mRNA levels, and genomic organization. J. Cell Biol. 94, 201–206
Chang, C., Meyerowitz, E.M. (1986) Molecular cloning and DNA sequence of the Arabidopsis thaliana alcohol dehydrogenase gene. Proc. Natl. Acad. Sci. USA 83, 1408–1412
Crouch, M.L. (1982) Non-zygotic embryos of Brassica napus L. contain embryo-specific storage proteins. Planta 156, 520–524
Crouch, M.L., Sussex, I.M. (1981) Development and storageprotein synthesis in Brassica napus L. embryos in vivo and in vitro. Planta 153, 64–74
Crouch, M.L., Tenbarge, K.M., Simon, A.E., Ferl, R. (1983) cDNA clones for Brassica napus seed storage proteins: Evidence from nucleotide sequence analysis that both subunits of napin are cleaved from a precursor polypeptide. J. Mol. Appl. Genet. 2, 273–283
Crouch, M.L., Tenbarge, K., Simon, A., Finkelstein, R., Scofield, S., Solberg, L. (1985) Storage protein mRNA levels can be regulated by abscisic acid in Brassica embryos. In: Molecular form and function of the plant genome, pp. 555–566, van Vloten-Doting, L., Groot, G.S.P., Hall, T.C., eds. Plenum Press, New York
Estelle, M., Somerville, C.R. (1986) The mutants of Arabidopsis. Trends in Genet. 2, 89–93
Finkelstein, R.R., Crouch, M.L. (1984) Precociously germinating rapeseed embryos retain characteristics of embryogeny. Planta 162, 125–131
Finlayson, A.J. (1976) The seed protein contents of some Cruciferae. In: The biology and chemistry of the Cruciferae, pp. 279–306, Vaughan, J.G., McLeod, A.J., Jones, B.M.G., eds. Academic Press, New York London
Forde, B.C., Kreis, M., Williamson, M.S., Fry, R.P., Pywell, J., Shewry, P.R., Bunce, N., Miflin, B.J. (1985) Short tandem repeats shared by B- and C-hordein cDNAs suggest a common evolutionary origin for two groups of cereal storage protein genes. EMBO J. 4, 9–15
Glossmann, H., Neville, D.M. (1971) Glycoproteins of cell surfaces. A comparative study of three different cell surfaces of the rat. J. Biol. Chem. 246, 6339–6346
Goding, L.A., Bhatty, R.S., Finlayson, A.J. (1970) The characterization of the 12 S “globulin” from rapeseed and its glycoprotein content. Can. J. Biochem. 48, 1096–1103
Goldberg, R.B., Fischer, R.L., Harada, J.J., Jofuku, D., Okamura, J.K. (1983a) Organization of soybean seed protein genomes and their flanking regions. In: Structure and function of plant genomes, pp. 37–45, Ciferri, O., Dure, L., eds. Plenum Press, New York
Goldberg, R.B., Hoschek, G., Vodkin, L.O. (1983b) An insertion sequence blocks the expression of a soybean lectin gene. Cell 33, 465–475
Higgins, T.J.V. (1984) Synthesis and regulation of major proteins in seeds. Annu. Rev. Plant Physiol. 35, 191–221
Horowitz, J. (1985) A Phaseolus mutation results in a reduced level of lectin mRNA. Mol. Gen. Genet. 198, 482–485
Koornneef, M., van Eden, J., Hanhart, C.J., Stam, P., Braaksma, F.J., Feenstra, W.J. (1983) Linkage map of Arabidopsis thaliana. J. Hered. 74, 265–272
Kreis, M., Shewry, P.R., Forde, B.G., Rahman, S., Miflin, B.J. (1983) Molecular analysis of a mutation conferring the highlysine phenotype on the grain of barley (Hordeum vulgare). Cell 34, 161–167
Ladin, B.F., Doyle, J.J., Beachy, R.N. (1984) Molecular characterization of a deletion mutation affecting the α-subunit of β-conglycinin of soybean. J. Mol. Appl. Genet. 2, 372–380
Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685
Laroche, M., Aspart, L., Delseny, M., Penon, P. (1984) Characterization of radish (Raphanus sativus) storage proteins. Plant Physiol. 74, 487–493
Leutwiler, L.S., Hough-Evans, B.R., Meyerowitz, E.M. (1984) The DNA of Arabidopsis thaliana. Mol. Gen. Genet. 194, 15–23
Lonnerdal, B., Janson, J.C. (1972) Studies on Brassica seed proteins. I. The low molecular weight proteins in rapeseed. Isolation and characterization. Biochim. Biophys. Acta 278, 175–183
Marsden, M.P.F., Meinke, D.W. (1985) Abnormal development of the suspensor in an embryo-lethal mutant of Arabidopsis thaliana. Am. J. Bot. 72, 1801–1812
Meinke, D.W. (1982) Embryo-lethal mutants of Arabidopsis thaliana: Evidence for gametophytic expression of the mutant genes. Theor. Appl. Genet. 63, 381–386
Meinke, D.W. (1985) Embryo-lethal mutants of Arabidopsis thaliana: Analysis of mutants with a wide range of lethal phases. Theor. Appl. Genet. 69, 543–552
Meinke, D.W. (1986) Embryo-lethal mutants and the study of plant embryo development. Oxford Surveys of Plant Molecular and Cell Biology 3, (in press)
Meinke, D.W., Baus, A.D. (1985) Gametophytic expression in embryo-lethal mutants of Arabidopsis thaliana. In: Biotechnology and ecology of pollen, pp. 15–20, Mulcahy, D., Mulcahy, G.B., Ottaviano, E., eds. Springer-Verlag, New York Berlin Heidelberg
Meinke, D.W., Chen, J., Beachy, R.N. (1981) Expression of storage-protein genes during soybean seed development. Planta 153 130–139
Meinke, D.W., Franzmann, L., Baus, A., Patton, D., Weldon, R., Heath, J.D., Monnot, C. (1985) Embryo-lethal mutants of Arabidopsis thaliana. In: Plant genetics, UCLA Symp. on Molec. and Cell. Biol. vol. 35, pp. 129–146, Freeling, M., ed. Alan Liss, New York
Meinke, D.W., Sussex, I.M. (1979a) Embryo-lethal mutants of Arabidopsis thaliana: A model system for genetic analysis of plant embryo development. Dev. Biol. 72, 50–61
Meinke, D.W., Sussex, I.M. (1979b) Isolation and characterization of six embryo-lethal mutants of Arabidopsis thaliana. Dev. Biol. 72, 62–72
Meyerowitz, E.M., Pruitt, R.E. (1985) Arabidopsis thaliana and plant molecular genetics. Science 229, 1214–1218
Müller, A.J. (1963) Embryonentest zum Nachweis rezessiver Letalfaktoren bei Arabidopsis thaliana. Biol. Zentralbl. 82, 133–163
Murashige, T., Skoog, F. (1962) A revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol. Plant. 15, 473–497
Pedersen, K., Devreux, J., Wilson, D.R., Sheldon, E., Larkins, B.A. (1982) Cloning and sequence analysis reveal structural variation among related zein genes in maize. Cell 29, 1015–1026
Rédei, G.P. (1975) Arabidopsis as a genetic tool. Annu. Rev. Genet. 9, 111–127
Schwenke, K.D., Raab, B., Linow, K.J., Pahtz, W., Uhlig, J. (1981) Isolation of the 12 S globulin from rapeseed (Brassica napus L.) and characterization as a “neutral” protein. Nahrung 25, 271–280
Schwenke, K.D., Raab, B., Plietz, P., Damaschun, G. (1983) The structure of the 12 S globulin from rapeseed (Brassica napus L.). Nahrung 27, 165–175
Simon, A.E., Tenbarge, K.M., Scofield, S., Finkelstein, R.R., Crouch, M.L. (1985) Nucleotide sequence of a cDNA clone of Brassica napus 12 S storage protein shows homology with legumin from Pisum sativum. Plant Mol. Biol. 5, 191–201
Somerville, C.R., McCourt, P., Caspar, T., Estelle, M., Keith, K. (1985) Arabidopsis thaliana as a model system for plant genetics and molecular biology. In: Plant genetics, UCLA Symp. on Molec. and Cell. Biol., vol. 35, pp. 651–660, Freeling, M., ed. Alan Liss, New York
Stuart, D.A., Nelsen, J. (1985) Factors affecting developmental processes in alfalfa cell cultures. In: Tissue culture in forestry and agriculture, pp. 59–73, Henke, R.R., Hughes, K.W., eds. Plenum Press, New York
Thomson, J.A., Schroeder, H.E. (1978) Cotyledonary storage proteins in Pisum sativum. II. Hereditary variation in components of the legumin and vicilin fractions. Aust. J. Plant Physiol. 5, 281–294
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Heath, J.D., Weldon, R., Monnot, C. et al. Analysis of storage proteins in normal and aborted seeds from embryo-lethal mutants of Arabidopsis thaliana . Planta 169, 304–312 (1986). https://doi.org/10.1007/BF00392124
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00392124