Abstract
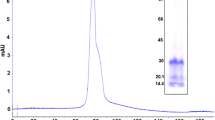
The occurrence of high haemagglutinating (lectin) activity in phloem exudate from three cucurbit species is reported. The protein responsible for this lectin activity in Cucurbita maxima Duch. has been isolated by cation exchange chromatography on Sepharose and identified by gel electrophoresis. The lectin showed agglutinating activity at concentrations as low as 0.1 μg/ml. No sugar, including those transported in the phloem of these species, interacted with agglutination. The lectin could not be extracted from cucurbit seed, but appeared in 5-day old seedlings. The possible role of a lectin in the sieve element is discussed.
Similar content being viewed by others
References
Albersheim, P., Anderson, A.J.: Proteins from plant cell walls inhibit polygalacturonases secreted by plant pathogens. Proc. Natl. Acad. Sci. USA 68, 1815–1819 (1971)
Albersheim, P., Jones, T.M., English, P.D.: Biochemistry of the cell wall in relation to infective processes. Ann. Rev. Phytopathol. 7, 171–194 (1969)
Allen, A.K., Neuberger, A., Sharon, N.: The purification, composition and specificity of wheat germ agglutinin. Biochem. J. 131, 155–162 (1973)
Anderson, A.J., Albersheim, P.: Host pathogen interactions V. Comparison of the abilities of proteins isolated from 3 varieties of Phaseolus vulgaris to inhibit the polygalacturonases secreted by three caces of Collectotrichum lindemuthianum. Physiol. Plant Pathol. 2, 339–346 (1972)
Beyenbach, J., Weber, C., Kleinig, H.: Sieve tube proteins from Cucurbita maxima. Planta 119, 113–124 (1974)
Burger, M.M.: Assays for agglutination with lectins. In: Methods in enzymology, pp. 615–621. Vol. 32, Part B. Fleischer, S., Packer, L., eds. New York: Academic Press 1974
Callow, J.A.: Plant lectins. In: Commentaries in plant science, pp. 221–233. Smith, H., ed. Oxford: Pergamon Press 1976
Cronshaw, J.: P-proteins. In: Phloem Transport, pp. 79–115. Aronoff, S., Dainty, J., Gorham, P.R., Srivastava, L.M., Swanson, C.A., eds. New York-London: Plenum Press 1975
Eschrich, W., Evert, R.F., Heyser, W.: Proteins of the sieve tube exudate of Cucurbita maxima. Planta 100, 208–221 (1971)
Evert, R.F., Eschrich, W., Eichorn, S.E.: P-protein distribution in mature sieve elements of Cucurbita maxima. Planta 109, 193–210 (1973)
Hamblin, J., Kent, S.P.: Possible role of phytohaemagglutinin in Phaseolus vulgaris L. Nature-New Biol. 245, 28–30 (1973)
Kauss, H., Ziegler, H.: Carbohydrate-binding proteins from the sieve-tube sap of Robinia pseudoacacia L. Planta 121, 197–200 (1974)
Kleinig, H., Thones, J., Dorr, I., Kollmann, R.: Filament formation in vitro of a sieve tube protein from Cucurbita maxima and Cucurbita pepo. Planta 127, 163–170 (1975)
Kollmann, R., Dorr, I., Kleinig, H.: Protein filaments—structural components of the phloem exudate. I. Observations with Cucurbita and Nicotiana. Planta 95, 86–94 (1970)
Liener, I.E.: Phytohemagglutinins (Phytolectins). Ann. Rev. Plant Physiol. 27, 291–319 (1976)
Lis, H., Sharon, N.: The biochemistry of plant lectins (phytohemagglutinins). Ann. Rev. Biochem. 42, 541–574 (1973)
Lowry, D.H., Rosebrough, N.J., Farr, N.L., Randall, R.L.: Protein measurement with the Folin phenol reagent. J. biol. Chem. 193, 265–275 (1951)
Matsson, O., Knox, R.B., Heslop-Harrison, J., Heslop-Harrison, Y.: Protein pellicle of stigmatic papillae as probable recognition site in incompatibility reactions. Nature 247, 298–300 (1974)
Mirelman, D., Galun, E., Sharon, N., Lotan, R.: Inhibition of fungal growth by wheat germ agglutinin. Nature 256, 414–416 (1975)
Nagata, Y., Burger, M.M.: Wheat germ agglutinin. Molecular characteristics and specificity for sugar binding. J. biol. Chem. 249, 3116–3122 (1974)
Reisfeld, R.A., Borjeson, J., Chessin, L.N., Small, P.A.: Isolation and characterisation of a mitogen from pokeweed (Phytolacca americana). Proc. Natl. Acad. Sci. USA 58, 2020–2027 (1967)
Rosen, S.D., Kafka, J.A., Simpson, D.L., Barondes, S.H.: Developmentally regulated carbohydrate-binding protein in Dictyostelium discoideum. Proc. Natl. Acad. Sci. USA 70, 2554–2557 (1973)
Ross, E., Schatz, G.: Assay of protein in the presence of high concentrations of sulfhydryl compounds. Anal. Biochem. 54, 304–306 (1973)
Sabnis, D.D., Hart, J.W.: A comparative analysis of phloem exudate proteins from Cucumis melo, Cucumis sativus and Cucurbita maxima by polyacrylamide gel electrophoresis and isoelectric focusing. Planta 130, 211–218 (1976)
Sharon, N., Lis, H.: Lectins: cell-agglutinating and sugarspecific proteins. Science 177, 949–959 (1972)
Sloan, R.T., Sabnis, D.D., Hart, J.W.: The heterogeneity of phloem exudate proteins from different plants: a comparative survey of ten plants using polyacrylamide gel electrophoresis. Planta 132, 97–102 (1976)
Vesterberg, O.: Isoelectric focusing of proteins in polyacrylamide gels. Biochim. Biophys. Acta 257, 11–19 (1972)
Walker, T.S.: The purification and some properties of a protein causing gelling in phloem sieve tube exudate from Cucurbita pepo. Biochim. Biophys. Acta 257, 433–444 (1972)
Waxdal, M.J.: Isolation, characterization and biological activities of five mitogens from pokeweed. Biochemistry 13, 3671–3677 (1974)
Webb, J.A., Gorham, P.R.: Translocation of photosynthetically assimilated C14 in straight-necked squash. Plant Physiol. 39, 663–672 (1964)
Weber, C., Franke, W.W., Kartenbeck, J.: Structure and biochemistry of phloem-proteins isolated from Cucurbita maxima. Exp. Cell Res. 87, 79–106 (1974)
Weber, K., Osborn, M.: The reliability of molecular weight determinations by dodecyl sulfate—polyacrylamide gel electrophoresis. J. biol. Chem. 244, 4406–4412 (1969)
Weise, L., Hayward, P.C.: On sexual agglutination and mating-type substances in isogamous dieocious Chlamydomonas. III. The sensitivity of sex cell contact to various enzymes. Am. J. Bot. 59, 530–536 (1972)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sabnis, D.D., Hart, J.W. The isolation and some properties of a lectin (Haemagglutinin) from Cucurbita phloem exudate. Planta 142, 97–101 (1978). https://doi.org/10.1007/BF00385126
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00385126