Abstract
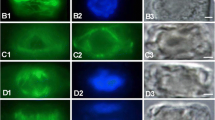
Protoplasts of the filamentous green alga Mougeotia sp. are spherical when isolated and revert to their normal cylindrical cell shape during regeneration of a cell wall. Sections of protoplasts show that cortical microtubules are present at all times but examination of osmotically ruptured protoplasts by negative staining shows that the microtubules are initially free and become progressively cross-bridged to the plasma membrane during the first 3 h of protoplast culture. Cell-wall microfibrils areoobserved within 60 min when protoplasts are returned to growth medium; deposition of microfibrils that is predominantly transverse to the future axis of elongation is detectable after about 6 h of culture. When regenerating protoplasts are treated with either colchicine or isopropyl-N-phenyl carbamate, drugs which interfere with microtubule polymerization, they remain spherical and develop cell walls in which the microfibrils are randomly oriented.
Similar content being viewed by others
References
Bouck, G.B., Brown, D.C.: Microtubule biogenesis and cell shape in Ochromonas 1. The distribution of cytoplasmic and mitotic microtubules. J. Cell Biol. 57, 340–359 (1973)
Burgess, J., Linstead, P.J.: Coumarin inhibition of microfibril formation at the surface of cultured protoplasts. Planta 133, 267–273 (1977)
Clarke, M., Schatten, G., Mazia, D., Spudich, J.A.: Visualization of actin fibers associated with the cell membrane in amoebae of Dictyostelium discoideum. Proc. Nat. Acad. Sci. USA 72, 1758–1762 (1975)
Coss, R.A., Bloodgood, R.A., Brower, D.L., Pickett-Heaps, J.D., McIntosh, J.R.: Studies on the mechanism of action of isopropyl N-phenyl carbamate. Exp. Cell Res. 92, 394–398 (1975)
Foos, K.: Mikrotubuli bei Mougeotia spec. Z. Pflanzenphysiol 62, 201–203 (1970)
Fowke, L.C., Pickett-Heaps, J.D.: Cell division in Spirogyra. I. Mitosis. J. Phycol. 5, 240–259 (1969)
Green, P.B., Erickson, R.D., Richmond, P.A.: On the physical basis of cell morphogenesis. Ann. N.Y. Acad. Sci. 175, 712–731 (1970)
Gunning, B.E.S., Hughes, J.E.: Quantitative assessment of symplastic transport of pre-nectar into the trichomes of Abutilon nectaries. Aust. J. Plant Physiol. 3, 619–637 (1976)
Hara, M., Umetsu, N., Miyamoto, C., Tamari, K.: Inhibition of the biosynthesis of plant cell wall materials, especially cellulose biosynthesis, by coumarin. Plant Cell Physiol. 14, 11–28 (1973)
Heath, I.B.: A unified hypothesis for the role of membrane bound enzyme complexes and microtubules in plant cell wall synthesis. J. Theor. Biol. 48, 445–449 (1974)
Hepler, P.K., Palevitz, B.A.: Microtubules and microfilaments. Ann. Rev. Plant Physiol. 25, 309–362 (1974)
Hogetsu, T., Shibaoka, H.: The change of pattern in microfibril arrangement on the inner surface of the cell wall of Closterium acerosum during cell growth. Planta 140, 7–14 (1978a)
Hogetsu, T., Shibaoka, H.: Effects of colchicine on cell shape and on microfibril arrangement in the cell wall of Closterium acerosum. Planta 140, 15–18 (1978b)
Hughes, J., McCully, M.E.: The use of an optical brightener in the study of plant structure. Stain Technol. 50, 319–329 (1975)
Itoh, T.: Microscopic and submicroscopic observation of the effects of coumarin and colchicine during elongation of pine seedlings. Plant Cell Physiol. 17, 367–384 (1976)
Koehler, J.K.: The freeze-etching technique. In: Principles and Techniques of Electron Microscopy. Biological Applications, vol. 2, pp. 51–98, Hayat, M.A., ed. New York, Cincinnati, Toronto, London, Melbourne: Van Nostrand Reinhold 1972
Marchant, H.J.: Microtubules associated with the plasma membrane isolated from protoplasts of the green alga Mougeotia. Exp. Cell Res. 115, 25–30 (1978a)
Marchant, H.J.: Scanning electron microscopy of microtubules associated with the plasma membrane of the alga Mougeotia. In: Scanning Electron Microscopy, vol 2, pp. 1071–1076, Becker, R.P., Johari, O., eds. O'Hare, Ill., USA: Scanning Electron Microscopy 1978b
Marchant, H.J.: Microtubules, cell wall deposition and the determination of plant cell shape. Nature 278, 167–168 (1979)
Marchant, H.J., Fowke, L.C.: Preparation, culture and regeneration of protoplasts from filamentous green algae. Can. J. Bot. 55, 3080–3086 (1977)
Marchant, H.J., Pickett-Heaps, J.D.: The effect of colchicine on colony formation in the algae Hydrodictyon, Pediastrum and Sorastrum. Planta 116, 291–300 (1974)
O'Brien, T.P.: The cytology of cell-wall formation in some eukaryotic cells. Bot. Rev. 38, 87–118 (1972)
Palevitz, B.A., Hepler, P.K.: Cellulose microfibril orientation and cell shaping in developing guard cells of Allium: the role of microtubules and ion accumulation. Planta 132, 71–93 (1976)
Robers, K.: Cytoplasmic microtubules and their functions. Prog. Biophys. Mol. Biol. 28, 373–420 (1974)
Robinson, D.G.: Structure, synthesis and the orientation of microfibrils. IV. Microtubules and microfibrils of Glaucocystis. Cytobiologie 15, 475–484 (1977)
Robinson, D.G., Grimm, I., Sachs, H.: Colchicine and microfibril orientation. Protoplasma 89, 375–380 (1976)
Schnepf, E., Röderer, G., Herth, W.: The formation of fibrils in the lorica of Poteriochromonas stipitata: tip growth, kinetics, site, orientation. Planta 125, 45–62 (1975)
Spurr, A.R.: A low viscosity epoxy embedding medium for electron microscopy. J. Ultrastruct. Res. 26, 31–43 (1969)
Wunderlich, F., Muller, R., Speth, V.: Direct evidence for a colchicine-induced impairment in the mobility of membrane components Science 182, 1136–1138 (1973)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marchant, H.J., Hines, E.R. The role of microtubules and cell-wall deposition in elongation of regenerating protoplasts of Mougeotia . Planta 146, 41–48 (1979). https://doi.org/10.1007/BF00381253
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00381253