Summary
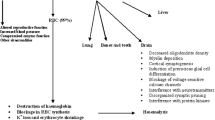
We have studied the effect of lead on the fluidity of erythrocyte membrane to clarify if lead can interact in vivo with biological membranes. Erythrocyte membranes were chosen in our study because a decrease of red cell osmotic fragility is also evident in the absence of laboratory and clinical signs of anaemia. The study was undertaken using the Electron Spin Resonance technique with two spin labels 5-doxyl stearate and 16-doxyl-stearate, which probe the physical state of the polar surface and the inner core of the membrane respectively.
Red blood cells and erythrocyte ghosts were prepared from the blood of workers occupationally exposed to lead and from healthy controls. The determinations of Pb blood, Pb urine, urine coproporphyrin and δ-amino levulinic acid showed an increased internal dose of lead, but the ordinary metabolic and haematological parameters were in the normal range. Our results show that in lead workers there is a change in chemical physical state both in erythrocytes and erythrocyte ghosts consistent with a decrease of membrane fluidity, which is evident in the surface as well as in the inner core of the membrane. The degree of membrane fluidity modification does not appear correlated with blood lead level. Changes in the membrane structural organization could be the molecular basis of some pathological alterations induced by lead.
Similar content being viewed by others
References
Aub JC, Reznikoff P (1924) The effects of lead in red blood cells. J Exp Med 40:189–193
Baker EL, Jr, Landigran PJ, Barbour AG, Cox DH, Folland DS, Ligo RN, Throckmorton J (1979) Occupational lead poisoning in the United States: clinical and biochemical findings related to blood levels. Br J Ind Med 36:314–322
Caspers ML, Siegel GJ (1980) Inhibition by lead of human erythrocyte (Na++K+)-adenosine triphosphate associated with binding of 210Pb to membrane fragments. Biochim Biophys Acta 600:27–35
Chapman D (1973) Physical chemistry of phospholipids. In: Ansell GB, Hawthorne JN, Dawson RMC (eds) Form and function of phospholipids. Elsevier Scientific Publishing Co., Amsterdam, pp 117–142
Clarkson TW, Kench JE (1958) Uptake of lead by human erythrocytes in vitro. Biochem J 69:432–434
Dacie JV, Lewis SM (1975) Practical haematology. Fifth Edition. Churchill Livingstone, Edinburgh London New York, pp 202–208
Davies JR, Andelman SC (1967) Urinary δ-aminolevulinic acid (ALA) levels in lead poisoning. I — A modified method for the rapid determination of urinary δ-aminolevulinic acid using disposable ion-exchange chromatography columns. Arch Environ Health 15:53–59
Eletr S, Inesi G (1972) Phase changes in the lipid moieties of sarcoplasmic reticulum membranes induced by temperature and protein conformational changes. Biochim Biophys Acta 290:178–185
Fukumoto K, Karai I, Horiguchi S (1980) The effect of lead on osmotic resistance of red blood cells. Proceedings of 53rd annual meeting of Japan Association of Industrial Health. May 15–17. Sendai, Japan, pp 533–534
Hessel DW (1968) A simple and rapid quantitative determination of lead in blood. Atomic Absorption Newsletter 7:55–56
Karai I, Fukumoto K, Horiguchi S (1981) Studies on osmotic fragility of red blood cells determined with a coil planet centrifuge for workers occupationally exposed to lead. Int Arch Occup Environ Health 48:273–281
Levine L, Fahy JP (1945) Evaluation of urinary lead determinations. I — The significance of the specific gravity. J Ind Hyg 27:217–223
Ong CN, Lee WR (1980) Distribution of lead-203 in human peripheral blood in vitro. Br J Ind Med 37:78–84
Rausa G, Calapaj GG (1970) Attività acetilcolinesterasica eritrocitaria e colinesterasica sierica di ratti trattati con piombo nitrato. Med Lav 61:554–562
Seelig J (1976) Spin labelling, theory and applications. Berlines LJ (ed). Academic Press, New York, pp 373–409
Selander S, Cramer K (1968) Determination of lead in urine by atomic absorption spectrophotometry. Br J Ind Med 25:139–143
Selhi HS, White JM (1975) The effect of lead on the red cell membrane. Postgrad Med J 51:765–769
Steck TL (1974) The organization of proteins in the human red blood cell membrane. J Cell Biol 62:1–21
Steck TL, Kant JA (1974) Preparation of impermeable ghosts and inside out vesicles from human erythrocyte membranes. Methods Enzymol 31:172–173
Talman EL, Schwartz S (1958) Porphyrins in urine. In: Seligson S, Standard methods of clinical chemistry 2:137
World Health Organization (1977) Environmental health criteria 3-Lead, WHO, Geneva
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valentino, M., Fiorini, R.M., Curatola, G. et al. Changes of membrane fluidity in erythrocytes of lead-exposed workers. Int. Arch Occup Environ Heath 51, 105–112 (1982). https://doi.org/10.1007/BF00378155
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00378155