Summary
A lectin with an affinity for β-d-galactoside-containing saccharides is present in the developing yolk sac from the chick embryo at stages from 2 to 7 days of incubation. This activity is present in the area vitellina (less differentiated) and the area vasculosa (more differentiated). In both areas, lectin activity increases significantly during the spreading of the yolk sac up to 5 days of incubation. At all of the stages studied lectin activity was significantly higher in the area vasculosa, as compared to the area vitellina.
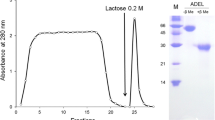
Lectins were purified by affinity chromatography and examined by SDS-PAGE. Under reducing conditions two components are evident. A more prominent band of subunit molecular weight of 14,200±100 for the area vitellina and 13,700±300 for the area vasculosa and a second band with molecular weight of about 68,000±700 and 68,000±1,200 for the area vitellina and area vasculosa respectively, were observed. The β-d-galactoside-binding lectin appears to be similar if not identical to that of the early chick blastoderm.
Similar content being viewed by others
References
Barondes SH (1984) Soluble lectins: a new class of extracellular proteins. Science 223:1259–1264
Boos W (1974) Pro and contra carrier proteins; sugar transport via the periplasmic galactose-binding protein. In: Bonner F, Kleinzeller A (eds) Current topics in membranes and transport, vol. 2. Academic Press, New York, pp 51–136
Boos W (1982) Synthesis of (2R)-glycerol-O-β-d-galactopyranoside by β-d-galactoside. Meth Enzymol 89:59–64
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cook GMW (1976) Techniques for the analysis of membrane carbohydrates. In: Maddy AH (ed) Biochemical analysis of membranes. Chapman and Hall, London, pp 283–351
Cook GMW, Zalik SE, Milos N, Scott V (1979) A lectin which binds specifically to β-d-galactoside groups is present at the earliest stages of chick embryo development. J Cell Sci 38:293–304
Cook GMW, Bellairs R, Rutherford NG, Stafford CA, Alderson T (1985) Isolation, characterization and localization of a lectin within the vitelline membrane of the hen's egg. J Embryol Exp Morphol 90:389–407
Dzandu JK, Deh ME, Barratt DL, Wise GE (1984). Detection of erythrocyte membrane proteins, sialoglycoproteins and lipids in the same polyacrylamide gel using a double-staining procedure. Proc Natl Acad Sci (USA) 81:1733–1737
Finney DJ, Hazelwood T, Smith MJ (1955) Logarithms to Base 2. J Gen Microbiol 12:222–225
Hamburger V, Hamilton HL (1951) A series of normal stages in the development of the chick embryo. J Morphol 88:49–92
Harrison FL, Chesterton CJ (1980a) Erythroid developmental agglutinin is a protein mediating specific cell-cell adhesion between differentiating rabbit erythroblasts. Nature (Lond) 286:502–504
Harrison FL, Chesterton CJ (1980b) Factors mediating cell recognition and adhesion. Galaptins, a recently discovered class of bridging molecules. FEBS Lett 122:157–165
Lis H, Sharon N (1981) Lectins in higher plants. In: Marcus A, Stumpf PK, Conn EE (eds) The biochemistry of plants, vol. 6. Academic Press, New York, pp 371–447
Milos N, Zalik SE (1982) Mechanisms of adhesion among cells of the early chick blastoderm. Role of the β-d-galactoside-binding lectin in the adhesion of extraembryonic endoderm cells. Differentiation 21:175–182
Milos N, Zalik SE (1986) Release of β-d-galactoside binding lectins into the cavities of aggregates of chick extraembryonic endoderm cells. Cell Differ 18:1–7
Nowak TP, Kobiler D, Roel LE, Barondes SH (1977) Developmentally regulated lectin from embryonic chick pectoral muscle. J Biol Chem 252:6026–6030
Phillips JR, Zalik SB (1982) Differential lectin-mediated agglutinabilities of the embryonic and the first extraembryonic cell line of the early chick embryo. Wilhelm Roux's Arch 191:234–240
Romanoff AL (1960) The avian embryo. Structural and functional development. MacMillan Co, New York
Weber K, Osborne M (1969) The reliability of molecular weight determinations by dodecyl sulphate-polyacrylamide gel electrophoresis. J Biol Chem 244:4406–4412
Wray W, Boulikas T, Wray VP, Hancock R (1981) Silver staining of proteins in polyacrylamide gels. Anal Biochem 118:197–203
Young MF, Minghetti PP, Klein NW (1980) Yolk sac endoderm: Exclusive site of serum protein synthesis in the early chick embryo. Dev Biol 75:239–245
Zalik SE, Cook GMW (1976) Comparison of embryonic and differentiating cell surfaces. Interactions of lectins with plasma membrane components. Biochim Biophys Acta 419:119–136
Zalik SE, Milos N (1986) Endogenous lectins and cell adhesion in embryonic cells. In: Browder L (ed) Developmental biology, a comprehensive synthesis, vol. II. Plenum Press, New York, pp 145–194
Zalik SE, Milos N, Ledsham I (1983) Distribution of two β-d-galactoside-binding lectins in the gastrulating chick embryo. Cell Differ 12:121–127
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mbamalu, G.M., Zalik, S.E. Endogenous β-d-galactoside binding lectin during the expansion of the yolk sac in the developing chick embryo. Roux's Arch Dev Biol 196, 176–184 (1987). https://doi.org/10.1007/BF00376312
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00376312