Abstract
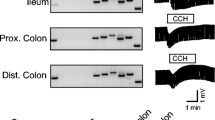
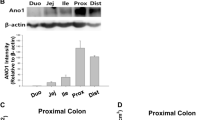
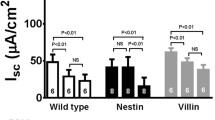
The cellular volume of crypts isolated from 2- to 3-week-old mouse small intestine has been measured to assess the capacity of the epithelial cells to respond to secretagogues. Vasoactive intestinal polypeptide (VIP) or carbachol, respectively cAMP- and calcium-mediated secretagogues, produced a reduction crypt volume attributed to KCl loss through channels activated by the agonists. Consistent with the participation of separate chloride channels, 4,4′-diisothiocyanatostilbene-2,2′-disulphonic acid (DIDS) blocked the carbacholbut not the VIP-induced volume decrease, whilst gl- ibenclamide abolished the VIP effect without affecting the carbachol-induced volume decrease. Animals homozygous for a disrupted cftr gene, introduced by gene targeting, were also used as the source for crypt isolation. In these CFTR(-/-) crypts, VIP failed to elicit any reduction in cellular volume, while the response to carbachol was indistinguishable from that seen in crypts from age-matched control animals. These results are consistent with murine CFTR being a cAMP-activated chloride channel inhibited by glibenclamide and resistant to DIDS. A separate chloride conductance activated by calcium mobilization in small-intestinal crypts appears to be independent of CFTR.
Similar content being viewed by others
References
Anderson MP, Welsh MJ (1991) Calcium and cAMO activate different chloride channels in the apical membrane of normal and cystic fibrosis epithelia. Proc Natl Acad Sci USA 88: 6003–6007
Anderson MP, Gregory RJ, Thompson S, Souza DW, Sucharita P, Mulligan RC, Smith AE, Welsh MJ (1991) Demonstration that CFTR is a chloride channel by alteration of its anion selectivity. Science 253: 202–205
Bear CE, Li C, Kartner N, Bridges RJ, Jensen TJ, Ramjeesingh M, Riordan JR (1992) Purification and functional reconstitution of the cystic fibrosis transmembrane conductance regulator (CFTR). Cell 68: 809–818
Berschneider HM, Knowles MR, Azizkhan RG, Boucher RC, Tobey NA, Orlando RC, Powell DW (1988) Altered intestinal chloride transport in cystic fibrosis. FASEB J 2: 2625–2629
Brown DR, Miller RJ (1992) Neurohormonal control of fluid and electrolyte transport in intestinal mucosa. In: Schultz SG (ed) Handbook of physiology — the gastrointestinal tract, vol IV. American Physiological Society, Bethesda, Maryland, pp 527–589
Clarke LL, Grubb BR, Gabriel SE, Smithies O, Koller BH, Boucher RC (1992) Defective epithelial chloride transport in a gene-targeted mouse model of cystic fibrosis. Science 257: 1125–1128
Cooke HJ (1987) Neural and humoral regulation of small intestinal electrolyte transport. In: Johnson LR (ed) Physiology of the gastrointestinal tract. Raven Press, New York, pp 1307–1350
Cotton CU, Stutts MJ, Knowles MR, Gatzy JT, Boucher RC (1987) Abnormal apical cell membrane in cystic fibrosis respiratory epithelium. An in vitro electrophysiological analysis. J Clin Invest 79: 80–85
Dorin JR, Dickinson P, Altwon EWFW, Smith SN, Geddes DM, Stevenson BJ, Kimber WL, Fleming S, Clarke AR, Hooper ML, Anderson L, Beddington RSP, Porteous DJ (1992) Cystic fibrosis in the mouse by targeted insertional mutagenesis. Nature 359: 211–215
Fuller CM, Benos DJ (1992) CFTR! Am J Physiol 263: C267-C286
Kartner N, Hanrahan JW, Jensen TJ, Naismith AL, Sun SZ, Ackerley CA, Reyes EF, Tsui LC, Rommens JM, Bear CE, Riordan JR (1991) Expression of the cystic-fibrosis gene in nonepithelial invertebrate cells produces a regulated anion conductance. Cell 64: 681–691
O'Brien JA, Walters RJ, Valverde MA, Sepúlveda FV (1991) Regulatory volume decrease in small intestinal crypts is inhibited by K+ and Cl− channel blockers. Biochim Biophys Acta 1070: 501–504
O'Brien JA, Walters RJ, Sepülveda FV (1993) Regulatory volume increase after hypertonicity- or VIP-induced cell volume decrease in small intestinal crypts is dependent on Na+/K+/2Cl− cotransport. Pflügers Arch 423: 67–73
Ratcliff R, Evans MJ, Doran J, Wainwright BJ, Williamson R, Colledge WH (1992) Disruption of the cystic fibrosis transmembrane conductance regulator gene in embryonic stem cells by gene targeting. Transgenic Res 1: 177–181
Ratcliff R, Evans MJ, Cuthbert AW, MacVinish LJ, Foster D, Anderson JR, Colledge WH (1993) Production of a severe cystic fibrosis mutation in mice. Nat Gen 4, 1: 35–41
Riordan JR, Rommens JM, Kerem B, Alon N, Rofmahel R, Grzelczak Z, Zielenski J, Lok S, Plavsic N, Chou J, Drumm ML, Iannuzzi MC, Collins FS, Tsui L (1989) Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245: 1066–1073
Sheppard DN, Welsh MJ (1992) Effect of ATP-sensitive K+ channel regulators on cystic fibrosis transmembrane conductance regulator chloride currents. J Gen Physiol 100: 573–591
Snouwaert JB, Brigman KK, Latzour AM, Malouf NN, Boucher RC, Smithies O, Koller BH (1992) An animal model for cystic fibrosis made by gene targeting. Science 257: 1083–1088
Tabcharani JA, Low W, Elie D, Hanrahan JW (1990) Low-conductance chloride channel activated by cAMP in the epithelial cell line T84. FEBS Lett 270: 157–164
Taylor CJ, Baxter PS, Hardcastle PT (1988) Failure to induce secretion in jejunal biopsies from children with cystic fibrosis. Gut 29: 957–962
Trezise AEO, Buchwald M (1990) In vivo cell-specific expression of the cystic fibrosis transmembrane conductance regulator. Nature 353: 434–437
Trezise A, Romano P, Gill DR, Hyde SC, Sepúlveda FV, Buchwald M, Higgins CF (1992) Expression of the multidrug and cystic fibrosis genes is regulated in a complementary fashion. EMBO J 11: 4291–4303
Walters RJ, Sepúlveda FV (1991) A basolateral K+ conductance modulated by carbachol dominates the membrane potential of small intestinal crypts. Pflügers Arch 419: 537–539
Walters RJ, O'Brien JA, Valverde MA, Sepúlveda FV (1992) Membrane conductance and cell volume changes evolved by VIP and carbachol in small intestinal crypts. Pflügers Arch 421: 598–605
Widdicombe JH (1986) Cystic fibrosis and β-adrenergic response of airway epithelial cell cultures. Am J Physiol 251: R818-R822
Widdicombe JH, Welsh MJ, Finkbeiner WE (1985) Cystic fibrosis decreases the apical membrane chloride permeability of monolayers cultured from cells of tracheal epithelium. Proc Natl Acad Sci USA 82: 6167–6171
Willumsen NJ, Boucher RC (1989) Activation of an apical Cl− conductance by Ca2+ ionophores in cystic fibrosis airway epithelia. Am J Physiol 256: C226-C233
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Valverde, M.A., O'Brien, J.A., Sepúlveda, F.V. et al. Inactivation of the murine cftr gene abolishes cAMP-mediated but not Ca2+-mediated secretagogue-induced volume decrease in small-intestinal crypts. Pflugers Arch. 425, 434–438 (1993). https://doi.org/10.1007/BF00374869
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374869