Abstract
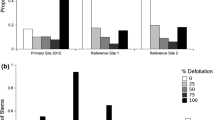
Sporadic patchy die-off of bush lupine, Lupinus arboreus, has long been known. We describe in detail a series of these incidents on the central California coast, based upon observational and comparative evidence. Stands of thousands of plants die, while nearby mature plants live on. In some sites, repeated die-off followed by regeneration from the seed bank has led to the cover and density of this woody, perennial plant fluctuating widely over the 40 year period for which records exist. Root damage by caterpillars of the ghost moth or “swift” Hepialus californicus (Lepidoptera, Hepialidae) is a major cause of individual bush death and a probable cause of die-off of stands of lupine. Hidden from view underground, a few of these insects readily kill a juvenile or young mature plant by girdling and reaming-out roots. The mass mortality of L. arboreus that we observed involved heavy root damage by these caterpillars in evenaged stands of plants in their first (1.5-year-old) or second (2.5-year-old) flowering season. The injured plants set seed before dying. Older, larger bush lupines better withstood root damage. In plants aged 3 or more years, damage and mortality were correlated with the intensity of ghost moth caterpillars in the roots. At the highest intensity (mean = 37.5, maximum = 62 caterpillars/root), a stand of large, old L. arboreus suffered 41% mortality; 45% of root cambium (median value) was destroyed by feeding caterpillars. Mass death of mature L. arboreus was not correlated with folivory, and leaf damage ranged from nil to moderate in instances of die-off. The western tussock moth, Orgyia vetusta, accounted for the highest levels of folivory, but this insect was rare when die-offs occurred. The lowest lupine mortality rates in our study occurred where tussock caterpillar intensities were high and where plants were repeatedly defoliated by this insect. However, experimental defoliation by high, but realistic, intensities of tussock moth caterpillars resulted in some mortality of mature bushes, and the combined effects of leaf and root herbivory have yet to be assessed. In its natural range on the California coast, bush lupine has several additional species of insect herbivores that can be locally abundant and injurious to the plant, although none is associated with die-off. Subterranean natural enemies of ghost moth caterpillars may play a role in the patchy waxing and waning of this shrub. Locally, a new species of entomophagous nematode (Heterorhabditis sp.) cause high mortality in the soil, before ghost moth caterpillars have entered the root. This natural enemy may thus afford lupines protection from heavy underground herbivory.
Similar content being viewed by others
References
Anderson DC (1987) Below-ground herbivory in natural communities: a review emphasizing fossorial animals. Quart Rev Biol 62:261–286
Barbour MG, Craig RB, Drysdale RR, Ghiselin MT (1973) Coastal ecology of Bodega Head. University of California Press, Berkeley
Bentley BL, Johnson ND (1991) Plants as food for herbivores: the roles of nitrogen fixation and carbon dioxide enrichment. In: Price PW, Fernandes GW, Lewinson TM, Benson WW (eds) Plant-animal interactions. Wiley, New York, pp 257–272
Brown VK, Gange AC (1990) Insect herbivory below ground. Adv Ecol Res 20:1–58
Crawley MJ (1989a) Insect herbivores and plant population dynamics. Annu Rev Ecol Syst 34:531–64
Crawley MJ (1989b) The relative importance of vertebrate and invertebrate herbivores in plant population dynamics. In: Bernays EA (ed) Insect-plant interactions, vol I. CRC Press, Boca Raton, pp 45–72
Davidson ED (1975) Demography of Lupinus arboreus at Bodega Head, CA. PhD dissertation, University of California, Davis
Davidson ED, Barbour MG (1977) Germination, establishment, and demography of coastal bush lupine at Bodega Head, California. Ecology 58:592–600
De Benedictis JA, Wagner DL, Whitfield JB (1990) Larval hosts of microlepidoptera of the San Bruno mountains, California. Atala 16:14–35
Esler AE (1974) Vegetation of the sand country bordering the Waitakere Range, Auckland: the southern beaches. Proc NZ Ecol Soc 21:72–77
Fritz RS, Simms EL (1992) Plant resistance to herbivores and pathogens: ecology, evolution, and genetics. University of Chicago Press, Chicago
Gagné RJ, Strong DR (1993) A new species of Dasineura (Diptera, Cecidomyiidae) galling the leaves of Lupinus spp (Fabaceae) in California. Proc Entomol Soc Wash 95:541–546
Hairston NG (1991) The literature glut: causes and consequences (reflections of a dinosaur). Bull Ecol Soc Am 72:171–174
Harrison S (1994) Resources and dispersal as factors limiting a population of the tussock moth (Orgyia vetusta), a flightless defoliator. Oecologia 99:27–34
Harrison S, Karban R (1986) Effects of an early-season folivorous moth on the success of a later-season species, mediated by a change in the quality of the shared host Lupinus arboreus Sims. Oecologia 69:354–359
Harrison S, Maron J (1995) Consequences of heavy insect herbivory for survival and seed output of bush lupine (Lupinus arboreus). Ecol Entomol (in press)
Louda S (1982) Limitation of the recruitment of the shrub Haplopappus squarrousus (Asteraceae) by flower and seed feeding insects. J Ecol 70:43–53
Louda SM, Potvin MA (1995) Effect of inflorescence-feeding insects on the demography and lifetime fitness of a native plant. Ecology 76:229–245
Margolis LR, Anderson RC, Holmes JC (1982) Recommended usage of selected terms in ecological and epidemiological parasitology. Bull Can Soc Zool 13:14
Molloy PJ, Partridge PR, Thomas WP (1991) Decline of tree lupin (Lupinus arboreus) on Kaitorete Spit, Canterbury, New Zealand, 1984–1990. NZ J Bot 29:349–352
Opler PA (1968) Unusual numbers of Hepialus sequoiolus Behrens in Sonoma County. PanPacific Entomol 44:83
Pitelka LP (1974) Energy allocation in annual and perennial lupines (Lupinus: Leguminosae). PhD dissertation, Stanford University, Palo Alto, CA, USA
Powell JA (1960) (abstract) PanPacific Entomol 36:45
Sih A (1991) Reflections on the power of a grand paradigm. Bull Ecol Soc Am 72:174–178
Stroyan HLG (1981) A North American lupin aphid found in Britain. Plant Pathol 30:253
Wagner DL (1995) Hepialid biology and behavior. Invertebr Taxon (in press)
Williams FX (1905) Notes on the life history of Hepilaus sequiolus Behrens. Entomol News 16:285
Williamson PA (1953) The ecology of the vegetation of sand dunes at Piha, Auckland. Rec Dominion Mus 2:19–35
Wink M (1992) The role of quinolizidine alkaloids in plant-insect interactions. In: Bernays E (ed) Plant-insect interactions, vol IV. CRC Press, Boca Raton, pp 131–166
Zandt HS (1994) A comparison of three sampling techniques to estimate the population size of caterpillars in trees. Oecologia 97:339–406
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Strong, D.R., Maron, J.L., Connors, P.G. et al. High mortality, fluctuation in numbers, and heavy subterranean insect herbivory in bush lupine, Lupinus arboreus . Oecologia 104, 85–92 (1995). https://doi.org/10.1007/BF00365566
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00365566