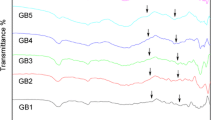
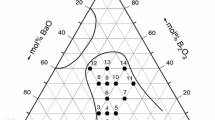
Abstract
Glasses were prepared with compositions (50−0.5 x) CaO.(50−0.5 x) P2O5 · x B2O3 with B2O3 contents (x) from 0 to 45 mol%. The glass transformation temperature (T g), dilatational softening temperature (T D) and Vickers hardness (H V) initially increased with x, but showed maxima at about x=20 for T g and T D and at about x=35 for H V. The thermal expansion coefficient decreased with x, levelling off at about 35 mol% B2O3. The maximum tendency to crystallize occurred at around 25 mol% B2O3. Volume nucleation (and hence glass-ceramic formation) and surface nucleation were obtained for x between 15 and 25 mol%. The first phase to appear was BPO4, which was probably homogeneously nucleated. Subsequently the 4CaO · P2O5 phase was heterogeneously nucleated on the BPO4. For 10⩾ x ⩾35 only surface nucleation was observed. The kinetics of nucleation were investigated in the 20 mol% B2O3 glass. The changes in properties and crystallisation behaviour with B2O3 content were related to short-range structural information. Infrared spectra and literature data indicated a threedimensional network of B-O-B and B-O-P linkages in the glasses.
Similar content being viewed by others
References
P. W. McMillan, “Glass Ceramics,” 2nd Edn (Academic, London, 1979).
P. F. James, in “Glasses and Glass Ceramics”, edited by M. H. Lewis (Chapman and Hall, London, 1989) pp. 59–105.
M. Akao, H. Aoki and K. Kato, J. Mater. Sci. 16 (1981) 809.
L. L. Hench, in Proceedings of 10th International Congress on Glass, Kyoto, July 1974 (Ceramic Society of Japan, Tokyo) No. 9, p. 30.
Y. Abe and H. Saito, J. Jpn. Ceram. Soc. 85 (1977) 45.
H. Bromer, K. Deutsher, B. Blenke, E. Pfeil and V. Strunz, Sci. Ceram. 9 (1977) 219.
T. Kokubo, S. Ito, M. Shigematsu, S. Sakka and T. Yamamuro, J.Mater. Sci. 20 (1985) 2001.
T. Nakamura, T. Yamamuro, S. Higashi, T. Kokubo and S. Ito, J. Biomed. Mater. Res. 19 (985) 685.
J. Vogel, W. Holand and W. Vogel, US Patent 4 698 318 (1987).
A. Osaka, Y. Toho, M. Asada and Y. Miura, Mater. Lett. 6 (1988) 347.
J. A. Wilder Jr, J. T. Healey and B. C. Bunker, in “Advances in Ceramics Vol 4”, edited by J. H. Simmons, D. R. Uhlmann and G. H. Beall (American Ceramic Society, Columbus, Ohio, 1982) pp. 313–333.
P. F. James, UK Patent 2 199 028 (1986).
T. H. Wang, PhD thesis, University of Sheffield (1987).
T. H. Wang and P. F. James, in “New Materials and their Applications” edited by D. Holland, Conference Series No. 111 (Institute of Physics, Bristol, 1990), p. 401.
R. T. Dehoff and F. N. Rhines, Trans. Metall. Soc. AIME 221 (1961) 975.
P. F. James, Phys. Chem. Glasses 15 (1974) 95.
I. Wozniak and P. F. James, Glass Technol. 25 (1984) 98.
J. Wong and C. A. Angell, “Glass Structure by Spectroscopy” (Dekker, New York, 1976) p. 864.
A. M. Abo-El-Azm, I. Kashif, H. Farouk, A. M. Sanad and Y. M. Abo-Zeid, Phys. Chem. Glasses 30 (1989) 251.
R. M. Almeida and J. D. Mackenzie, J. Non-Cryst. Solids 40 (1980) 535.
C. A. Angell, in Proceedings of 2nd International Symposium on Halide Glasses, Rensselaer Polytechnic Institute, Troy, New York, 1983, Paper No. 8 (extended abstract).
A. E. R. Westman, in “Modern Aspects of the Vitreous State I”, edited by J. D. Mackenzie (1960) p. 63.
J. C. Arriagada, W. Burckhardt and A. Felty, J. Non-Cryst. Solids 91 (1987) 375.
W. L. Konijnendijk and J. M. Stevels, in “Borate Glasses”, Materials Science Research Vol. 12, edited by L. D. Pye, V. D. Frechette and N. J. Kreidl (Plenum, New York, 1978) p. 259.
D. L. Griscom, ibid., p. 35.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shi, W., James, P.F. Crystallization and properties of CaO-P2O2-B2O3 glasses. JOURNAL OF MATERIALS SCIENCE 28, 469–476 (1993). https://doi.org/10.1007/BF00357825
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00357825