Summary
The number of flowers produced by inflorescences of Yucca whipplei (Agavaceae) consistently exceeds the number of fruits produced by about one order of magnitude. To determine the factors responsible for low fruit set, the relation between pollinator availability, the amount of resources spent on reproduction (as indicated by inflorescence size), and the number of fruits matured was studied during 1978 and 1979 at 18 locations in chaparral, coastal sage scrub, and desert scrub communities of southern California.
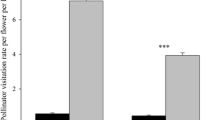
The following results support the conclusion that pollinators do not usually limit fruit production in Yucca whipplei. Rather, fruit production is limited by the amount of resources available to support developing fruits. (1) Fruit production is positively correlated with inflorescence size both within and between populations. The average size of inflorescence for a population is an excellent predictor of mean fruit production. Furthermore, 54% of the total variance in fruit production of individual plants can be explained by inflorescence size. (2) In contrast, although fruit production within most populations is positively correlated with an index of the number of pollinator visits to an inflorescence, the relative abundance of pollinators for a population is a poor predictor of mean fruit production, and only 9% of the total variance in fruit production can be explained by the visitation index. Furthermore, at four sites studied for two years, there was little change in average inflorescence size or fruit production from 1978 to 1979, despite large differences in relative abundance of pollinators at each of the sites. (3) Based on geographic proximity, and physiographic and vegetational similarities, study sites were grouped into regional clusters. Both inflorescence size and fruit production varied considerably between regions. Of the total variation in fruit production, 27% can be attributed to differences between regions. Most of this variation is the result of regional differences in inflorescence size, which in turn influence fruit production.
Why does Yucca whipplei produce such large inflorescences if so few fruits can be supported? Two relevant hypotheses are discussed: (1) the floral display is the result of selection for pollen dissemination at the expense of fruit set; and (2) the floral display is the result of selection for a bet-hedging strategy either to increase the probability of adequate pollination when pollinators are unusually rare, or to allow individuals to support more fruits when resources are unusually abundant.
Similar content being viewed by others
References
Aker C, Udovic D (1981) Oviposition and pollination behavior of the yucca moth Tegeticula maculata (Lepidoptera: Proxidae), and its relation to the reproductive biology of Yucca whipplei (Agavaceae). Oecologia, in press
Baker HG, Baker I (1975) Studies of nectar-constitution and pollinator-plant coevolution. In: LE Gilbert, PH Raven (eds) Coevolution of Animals and Plants. University of Texas Press, Austin, Texas, p 100–140
Brown JH, Kodric-Brown A (1979) Convergence, competition and mimicry in a temperate community of hummingbird pollinated flowers. Ecology 60:1022–1035
Charnov EL (1979) Simultaneous hermaphroditism and sexual selection. Proc Nat Acad Sci US 76:2480–2484
Faegri K, vander Pijl L (1971) The principles of pollination ecology, 2nd ed., pp 291, Pergamon Press, Oxford
Frankie GW (1975) Tropical forest phenology and pollinator plant coevolution. In: LE Gilbert, PH Raven (eds) Coevolution of Animals and Plants. University of Texas Press, Austin, Texas, p 192–209
Free JB (1970) Insect Pollination of Crops. Academic Press NY, pp 544
Goh BS (1979) Stability in models of mutualism. American Naturalist 113:261–275
Haines L (1941) Variation in Yucca whipplei. Madrono 6:33–45
Harrison AT, Small E, Mooney HA (1971) Drought relationships and distribution of two mediterranean-climate California plantcommunities. Ecology 52:869–875
Heinrich B, Raven PH (1972) Energetics and pollination ecology. Science 176:597–602
Heithaus ER (1974) The role of pollinator-plant interactions in determining community structure. Ann Mo Bot Garden 61:675–691
Lioyd DG (1979) Parental strategies of angiosperms. New Zealand J Bot 17:595–606
Lioyd DG (1980) Sexual strategies in plants. I. An hypothesis of serial adjustment of maternal investment during one reproductive session. New Phytologist, 86:69–79
May RM (1976) Models for two interacting populations. In: RM May (ed) Theoretical Ecology: Principles and Applications. Saunders, Philadelphia, PA, p 49–70
Maynard Smith J (1977) The sex habit in plants and animals. In: FB Christiansen, TM Fenchel (eds) Measuring Selection in Natural Populations. Springer Verlag, New York Berlin Heidelberg, p 315–331
McGregor SE (1959) Cotton-flower visitation and pollen distribution by honey bees. Science 129:97–98
McKelvie SD (1947) Yuccas of the Southwestern United States. Part II, Harvard Univ, Cambridge: Arnold Arboretum, pp 192
Mosquin T (1971) Competition for pollinators as a stimulus for the evolution of flowering time. Oikos 22:398–402
Munz PA (1973) A California Flora and Supplement. University of California Press, Berkeley, pp 1681–224
Nishida T (1963) Ecology of the pollinators of passion fruit. Tech Bull Hawaii Agric Exp Stn 55
Fowell JA, Mackie RA (1966) Biological interrelationships of moths and Yucca whipplei. Univ of Calif Publ in Entomol 42:1–59
Riley CV (1892) The yucca moth and Yucca pollination. Ann Rept Missouri Bot Garden 3:99–158
Sehaffer WM (1979) Equivalence of maximizing reproductive value und fitness in the case of reproductive strategies. Proc Nat Acad Sci US 76:3567–3569
Schaffer WM, Gadgil MD (1976) Selection for optimal life history strategies in plants. In: ML Coly, JM Diamond (eds) Ecology and Evolution of Communities. Belknap Press, Cambridge, MA, p 142–157
Schaffer WM, Schaffer MV (1977) The adaptive significance of variations in reproductive habit in Agavaceae. In: B Stonehouse (ed) Evolutionary Ecology. Univ Park Press, College Park MD, p 261–276
Schaffer WM, Schaffer MV (1979) The adaptive significance of variations in reproductive habit in the Agavaceae. II: Pollinator foraging behavior and selection for increased reproductive expenditure. Ecology 60:1051–1069
Schemske DW (1977) Flowering phenology and seed set in Claytonia virginica (Portulacaceae). Bull Torrey Botanical Club 104:254–263
Schemske DW (1980) The evolution of floral display in the orchid Brassavola nodosa. Evolution 34:489–494
Stephenson AG (1979) An evolutionary examination of the floral display of Catalpa speciosa (Bignoniaceae). Evolution 33:1200–1209
Trelease W (1893) Further studies of yuccas and their pollination. Ann Rept Missouri Bot Garden 4:181–226
Udovic D, Aker C 1981 Fruit abortion and the regulation of fruit number in Yucca whipplei. Oecologia, in press
Vandermeer JH, Boucher DH (1978) Varieties of mutualistic interaction in population models. Journal of Theoretical Biology 74:549–558
Vasek FC, Thorne RF (1977) Transmontane coniferous vegetation. In: MG Barbour, J Major (eds) Terrestrial vegetation of California. Wiley and Sons, New York, p 797–832
Waser NM (1978) Competition for pollination and sequential flowering in two Colorado wild flowers. Ecology 59:934–944
Webber JM (1953) Yuccas of the Southwest. US Dept Agric Monogr 17: pp 9
Willson MF (1979) Sexual selection in plants. American Naturalist 113:777–790
Willson MF, Price PW (1977) The evolution of inflorescence size in Asclepias (Asclepiadaceae). Evolution 31:495–511
Willson MF, Rathke B (1974) Adaptive design of the floral display in Asclepias syriaca L. American Midland Naturalist 92:47–57
Wimber DR (1958) Pollination of Yucca whipplei Torr. Masters thesis, Calremont College (unpublished)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Udovic, D. Determinants of fruit set in Yucca whipplei: Reproductive expenditure vs. pollinator availability. Oecologia 48, 389–399 (1981). https://doi.org/10.1007/BF00346500
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00346500