Summary
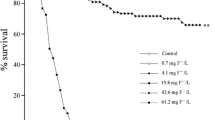
Tail fin pieces (“discs”) of Rana pipiens tadpoles were treated with thyroxine (T4) solution (50 μg/liter) and the induced changes were studied with the light and electron microscopes. Definitive effects first appeared after 4 days of treatment. Thereafter various resorptive processes proceeded at an accelerating pace. By the 9th or 10th day the discs were reduced to tiny spherules.
The most conspicuous changes were those of the breakdown and disposal of the basement lamella. First there was a “loosening” of the collagen layers followed by massive infiltration by macrophages which engulfed the collagen fibrils. By the 8th day the phagocytosis of the basement lamella was completed for the most part and the macrophages were clumped into masses.
After 6 days of T4 treatment the epidermis was transformed from a 2- or 3-cell layered epithelium to a multilayered one. This was due to the detachment of the basal epithelial cells from the basement lamella followed by their movement towards the surface. Later, the epidermal cells showed atrophy by extensive autolytic processes.
Discs incubated in the control medium (devoid of T4) remained intact for the duration of the experiment. Their morphology was essentially the same as that described by others from studies in vivo.
Similar content being viewed by others
References
Bennet, H. S.: Morphological aspects of intracellular polysaccharides. J. Histochem. Cytochem. 11, 14–23 (1963).
Berkowitz, L.: Staining of nervous tissue following extraction of araldite embedment including differential staining of glial elements and cell organelles. Anat. Rec. 160, 507 (1968).
Derby, A.: An in vitro quantitative analysis of the response of tadpole tissue to thyroxine. J. exp. Zool. 168, 147–156 (1968).
Edds, M. V., Jr., and P. R. Sweeny: Chemical and morphological differentiation of the basement lamella. In: Synthesis of molecular and cellular structure, 19th Growth Symposium, ed. by D. Rudnick, p. 111–138. New York: Ronald 1960.
—, and P. R. Sweeny: Development of the basement lamella. Fifth Int. Congr. El. Micr., Proceedings, vol. 2. QQ-2. New York: Academic Press 1962.
Eisen, A. Z., and J. Gross: The role of epithelium and mesenchyme in the production of collagenase and hyaluronidase in the anuran tadpole. Fed. Proc. 24, 558 (1965).
Etkin, W.: Growth and resorption phenomena in anuran metamorphosis. Physiol. Zool. 5, 275–300 (1932).
Frieden, E.: Biochemical adaptation in anuran metamorphosis. Amer. Zool. 1, 115–149 (1961).
Gudernatsch, J. F.: Feeding experiments on tadpoles. I. The influence of specific organs given as food on growth and differentiation. A contribution to the knowledge of organs with internal secretion. Wilhelm Roux' Arch. Entwickl.-Mech. Org. 35, 457–483 (1912).
Hay, E. D.: Electron microscopic observations of muscle dedifferentiation in regenerating Amblystoma limbs. Develop. Biol. 1, 555–585 (1959).
Helff, O. M.: Factors involved in the atrophy of the tail of anuran larvae during metamorphosis. Anat. Rec. 34, 129 (1926).
—: Studies on amphibian metamorphosis. X. Hydrogen-ion concentration of the blood of anuran larvae during involution. Biol. Bull. 63, 405–418 (1932).
Kaltenbach, J. C.: Local action of thyroxine on amphibian metamorphosis. J. exp. Zool. 140, 1–18 (1959).
Kelly, D. E.: Fine structure of desmosomes, hemidesmosomes, and an adepidermal globular layer in developing newt epidermis. J. Cell Biol. 68, 51–72 (1966).
Kemp, N. E.: Development of the basement lamella of larval anuran skin. Develop. Biol. 1, 459–476 (1959).
—: Replacement of the larval basement lamella by adult-type basement membrane in anuran skin during metamorphosis. Develop. Biol. 3, 391–410 (1961).
—: Metamorphic changes of dermis in skin of frog larvae exposed to thyroxine. Develop. Biol. 7, 244–254 (1963).
Metchnikoff, E.: Atrophie des muscles pendant la transformation des batraciens. Ann. Inst. Pasteur 6, 1–12 (1892).
Reynolds, E. S.: The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J. Cell Biol. 17, 208–212 (1963).
Rugh, R.: Induced ovulation and artificial fertilization in the frog. Biol. Bull. 66, 22–29 (1934).
Salpeter, M. M., and M. Singer: The fine structure of the adepidermal reticulum in the basal membrane of the skin of the newt, Triturus. J. biophys. biochem. Cytol. 6, 35–40 (1959).
—, and M. Singer: Differentiation of the submicroscopic adepidermal membrane during limb regeneration in adult Triturus, including a note on the use of the term basement membrane. Anat. Rec. 136, 27–40 (1960).
Scharrer, B.: Ultrastructural study of the regressing prothoracic glands of blattarian insects. Z. Zellforsch. 69, 1–21 (1966).
Usuku, G., and J. Gross: Morphologic studies of connective tissue resorption in the tail fin of metamorphosing bullfrog tadpole. Develop. Biol. 11, 352–370 (1965).
Weber, R.: Induced metamorphosis in isolated tails of Xenopus laevis. Experientia (Basel) 18, 84–85 (1962).
—: Ultrastructural changes in regressing tail muscles of Xenopus laevis at metamorphosis. J. Cell Biol. 22, 481–487 (1964).
Weiss, P., and W. Ferris: Electron-microscopic study of the texture of the basement membrane of larval amphibian skin. Proc. nat. Acad. Sci. (Wash.) 40, 528–540 (1954).
—, and W. Ferris: The basement lamella of amphibian skin: Its reconstruction after wounding. J. biophys. biochem. Cytol. 2, Suppl., 275–281 (1956).
Author information
Authors and Affiliations
Additional information
This study was supported by Grants NIH T1-GM-102, NSF GB-5913, NIH NB-00840 and NIH NB-07566, and an Institutional Grant from the New Jersey College of Medicine and Dentistry.
I am indebted to Drs. J. Osinchak and W. Etkin for their encouragement.
Rights and permissions
About this article
Cite this article
Gona, A.G. Light and electron microscopic study on thyroxine-induced in vitro resorption of the tadpole tail fin. Z. Zellforsch. 95, 483–494 (1969). https://doi.org/10.1007/BF00335142
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00335142