Summary
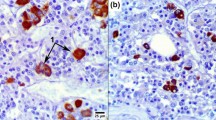
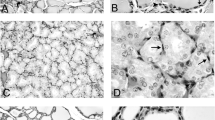
The characteristic ultrastructure of thyrotrophs of the rat anterior pituitary was observed by immuno-electron microscopy and enzyme cytochemistry with increasing time after thyroidectomy (TX). The rough endoplasmic reticulum (ER) became dilated, the intracisternal granules reacted to serum raised against thyroid stimulating hormone (TSH) around 21 days after TX, and lysosomes and peculiar structures with positive acid phosphatase activity were present. The administration of thyroxine (T4) to the thyroidectomized rats resulted in the reformation of secretory granules, a reduction of dilated cisternae of rough ER and the activation of the lysosomal systems. Morphological features indicating that the TX-cells might be derived from growth hormone (GH) cells or cells other than TSH cells, previously suggested by some researchers, were not recognized in the present study. The amount of serum and pituitary TSH was measured by radioimmunoassay (RIA), and correlated well with the morphological changes. These results indicate that the TX-cells are hypertrophied hyperfunctioning TSH cells that have been affected by the lack of negative feedback of thyroid hormone.
Similar content being viewed by others
References
Barka T, Anderson PJ (1962) Histochemical methods for acid phosphatase using hexazonium pararosanilin as coupler. J Histochem Cytochem 10:741–753
Contopoulos AN, Simpson ME, Koneff AA (1958) Pituitary function in the thyroidectomy rat. Endocrinology 63:642–653
Dingemans KP (1969) On the origin of thyroidectomy cells. J Ultrastruct Res 26:480–500
Elftman H (1958) Origin of thyroidectomy cell. Anat Rec 131:119–127
Farquhar MG (1971) Processing of secretory products by cells of the anterior pituitary gland. Mem Soc Endocrinol 19:79–124
Farqhar MG, Rinehart JF (1954) Cytologic alterations in the anterior pituitary gland following thyroidectomy: an electron microscope study. Endocrinology 55:857–876
Goluboff LG, Macrae ME, Ezrin C, Sellers EA (1970) Autoradiography of tritiated thymidine labeled anterior pituitary cells in propylthiouracil treated rats. Endocrinology 87:1113–1118
Kiguchi Y (1978) The process of development of thyroidectomy cells from the so-called thyrotrophs. Endocrinol Jpn 25:75–86
Kurosumi K, Baba N (1969) Experimental and histochemical studies on the rat pituitary thyrotrophs by electron microscopy. Gunma Symp Endocrinol 6:197–210
Kurosumi K, Inoue K (1986) Ultrastructure of anterior pituitary cells. Curr Top Neuroendocrinol 7:99–134
Kurosumi K, Oota Y (1966) Corticotrophs in the anterior pituitary glands of gonadectomized and thyroidectomized rats as revealed by electron microscopy. Endocrinology 79:808–814
Moriarty GC, Tobin RB (1976) An immunocytochemical study of TSH storage in rat thyroidectomy cells with and without D or L thyroxine treatment. J Histochem Cytochem 24:1140–1149
Okino H, Matsui S, Shioda S, Nakai Y, Kurosumi K (1979) Ultrastructural and morphometric studies on the rat pituitary and thyroid follicular cells following administration of thyrotropin releasing hormone. Arch Histol Jpn 42:489–505
Ozawa H, Kurosumi K (1989) Postnatal development of thyrotrophs in the rat anterior pituitary as studied by immunogold electron microscopy. Anat Embryol 180:207–212
Palade GE (1956) Intracisternal granules in the exocrine cells of the pancreas. J Biophys Biochem Cytol 2:417–422
Rambourg A (1967) An improved silver methanamine technique for the detection of periodic acid-reactive complex carbohydrates with the electron microscope. J Histochem Cytochem 15:409–412
Rosa GC, D'Angelo SA (1974) The ultrastructure of the thyrotropic cell during thyrotropin rebound in the adenohypophysis of the rat. Am J Anat 135:33–50
Schiaffino S, Hanzlíková V (1972) Autophagic degradation of glycogen in skeletal muscles of the newborn rat. J Cell Biol 52:41–51
Shiino M, Williams MG, Rennels EG (1972) Thyroidectomy cells and their response to thyrotropin releasing hormone (TRH) in the rat. Z Zellforsch 138:327–332
Smith R, Farquhar MG (1966) Lysosome function in the regulation of the secretory process in cells of the anterior pituitary gland. J Cell Biol 31:319–347
Stratmann IE, Ezrin C, Sellers EA, Simon GT (1972) The origin of the thyroidectomy cells as revealed by high resolution radioautography. Endocrinology 90:728–734
Tougard C, Picart R, Tixier-Vidal A (1982) Immunocytochemical localization of prolactin in the endoplasmic reticulum of GH3 cells. Variations in response to thyroliberin. Biol Cell 43:89–102
Wakabayashi K, Tanaka S (1988) Assessment of specificity of antisera for immunohistochemistry. Acta Histochem Cytochem 21:221–229
Yang HJ, Ozawa H, Kurosumi K (1989) Ultrastructural changes in growth hormone cells in the rat anterior pituitary after thyroidectomy. J Clin Electron Microsc 22:269–283
Yoshimura F, Harumiya K, Soji T, Yokoyama M, Kumagai T (1973) Possible reversion of pituitary “thyroidectomy cells” into their original acidophils in rats. Endocrinol Jpn 20:249–262
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ozawa, H. Changing ultrastructure of thyrotrophs in the rat anterior pituitary after thyroidectomy as studied by immuno-electron microscopy and enzyme cytochemistry. Cell Tissue Res 263, 405–412 (1991). https://doi.org/10.1007/BF00327274
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00327274