Summary
The pattern of metabolites of ciclosporin in blood and 24 h-urine of 58 liver graft recipients was routinely monitored by HPLC from transplantation until discharge from hospital. Liver function and ciclosporin metabolite pattern in patients with an uncomplicated clinical course and in those with cholestasis or acute rejection were compared.
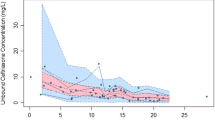
During cholestasis M19 and M1A, and during acute rejection M19, in blood were significantly elevated compared to the control group. Blood M19 was significantly correlated with bilirubin concentration and γ-glutamyl transferase activity in serum, and M1A with the serum bilirubin concentration. Analysis of the metabolite pattern over the observation period showed higher concentrations of M19 and M1A in blood from patients with cholestasis and acute rejection than in the control group; concentrations were lower in the rejection group than in the cholestasis group. The metabolite pattern in 24 h-urine showed similar alterations in ciclosporin metabolite pattern to those in blood.
Cholestasis and rejection shift the ciclosporin metabolite pattern in blood and urine to higher concentrations of M19 and M1A, whereas the concentrations of other metabolites and ciclosporin were not significantly affected.
Similar content being viewed by others
References
Combalbert J, Fabre J, Fabre G, Dalet I, Derancourt J, Cano JP, Maurel P (1989) Metabolism of cyclosporin A: IV. Purification and identification of the rifampicin-inducible human liver cytochrome P450 (cyclosporin A oxidase) as a product of P450IIIA gene subfamily. Drug Metab Dispos 17: 197–207
Wallemacq PE, Lhoëst G, Dumont P (1989) Isolation, purification and structure elucidation of cyclosporin metabolites in rabbit and man. Biomed Environ Mass Spectrom 18: 48–56
Wagner O, Schreier E, Heitz F, Maurer G (1987) Tissue distribution, disposition, and metabolism of cyclosporine in rats. Drug Metab Dispos 15: 377–383
Maurer G, Lemaire M (1986) Biotransformation of cyclosporine and distribution in blood. Transplant Proc 18: 25–34
Kohlhaw K, Wonigeit K, Schäfer O, Ringe B, Bunzendahl H, Pichlmayr R (1989) Association of very high blood levels of cyclosporine metabolites with clinical complications after liver transplantation. Transplant Proc 21: 2232–2233
Wonigeit K, Kohlhaw K, Winkler M, Schaefer O, Pichlmayr R (1990) Cyclosporine monitoring in liver allograft recipients. Two distinct patterns of blood level derangement associated with nephrotoxicity. Transplant Proc 22 (3): 1305–1311
Christians U, Zimmer KO, Wonigeit K, Maurer G, Sewing KF (1988) Liquid chromatographic measurement of cyclosporin A and its metabolites in blood, bile, and urine. Clin Chem 34: 34–39
Christians U, Sewing KF (1989) Whole blood sample clean-up for chromatographic analysis. In Zech K, Frei RW (eds) Selective sample handling and detection in high-performance liquid chromatography, part B. Elsevier, Amsterdam, pp 82–132
Christians U, Zimmer KO, Wonigeit K, Sewing KF (1987) Measurement of cyclosporin A and four of its metabolites in whole blood by high-performance liquid chromatography. J Chromatogr 413: 121–129
Kahan BD (1990) Consensus report of the ciclosporin expert meeting, Hawk's Kay, 1990. Transplant Proc 22 (3): 1357–1361
Wadhwa NK, Schroeder TJ, Amadeo JP, Myre SA, Clardy SW, First MR (1987) Cyclosporine drug interactions: a review. Ther Drug Monit 9: 399–406
Cockburn ITR, Krupp P (1989) An appraisal of drug interactions with Sandimmun®. Transplant Proc 21: 3845–38507
Jain AB, Elias E, Gunson BK, Buckels JAC, McDowell B, Hillis A, McMaster P (1988) Is elimination of cyclosporine independent on liver graft function and biliary drainage? Transplant Proc 20: 516–522
Sommerauer JF, Grant D, Freeman D, Mimeaul R, Duff J, Wall W (1989) Effects of cholestasis on cyclosporine metabolism in the pig. Transplant Proc 21: 835–836
Oldhafer KJ, Schumann G, Wonigeit K, Oellerich M, Ringe B, Pichlmayr R (1988) Cyclosporine A monitoring by radioimmunoassay (RIA) and high-performance liquid chromatography (HPLC) after liver transplantation: Influence of route of administration and of liver function on the RIA: HPLC ratio. Transplant Proc 20: 361–365
Burckart G, Starzl T, Williams L, Sanghvi A, Gartner C, Venkataramanan R, Zitelli B, Malatack J, Urbach A, Diven W, Ptachcinski R, Shaw B, Iwatsuki S (1985) Cyclosporine monitoring and pharmacokinetics in pedriatic liver transplant patients. Transplant Proc 17: 1172–1175
Bleck JS, Schlitt HJ, Christians U, Thiesemann C, Strohmeyer S, Schottmann R, Kohlhaw K, Wonigeit K, Sewing KF (1989) Urinary excretion of ciclosporin and 17 of its metabolites in renal allograft recipients. Pharmacol 39: 160–164
Christians U, Schlitt HJ, Bleck JS, Schiebel HM, Kownatzki R, Maurer G, Strohmeyer S, Schottmann R, Wonigeit K, Pichlmayr R, Sewing KF (1988) Measurement of ciclosporin and 18 metabolites in blood, bile and urine by high-performance liquid chromatography (HPLC). Transplant Proc 20: 609–613
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Christians, U., Kohlhaw, K., Budniak, J. et al. Ciclosporin metabolite pattern in blood and urine of liver graft recipients. Eur J Clin Pharmacol 41, 291–296 (1991). https://doi.org/10.1007/BF00314954
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00314954