Summary
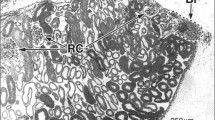
The cytoarchitecture of the interstitial tissue of the rat kidney was studied by combined scanning and transmission electron microscopy. The renal interstitium is composed of an elaborate network of stellate sustentacular cells. In the cortex, sustentacular cells radiate thin branching processes to form a fine reticulum, which supports intertubular spaces. In the medulla, these cells extend thick processes horizontally along the basal surfaces of the thin limbs or vasa recta, reinforcing their attenuate walls. The horizontal processes connect with each other at their terminals, compartmentalizing the interstitial space into thin layers. The medullary sustentacular cells contain abundant small lipid droplets. The network of sustentacular cells houses vasa recta, keeping them in parallel position to each other and to the tubules. The arterial vasa recta are accompanied by pericytes, which frequently contain lipid droplets larger in size than those in the sustentacular cells. Venous vasa recta extend numerous basal microvilli, which anchor the venous wall to adjacent tubules or vessels. Numerous free cells, round in shape, are found in the sustentacular cell network, especially in the cortex. They consist of macrophages and occasional lymphocytes. Some macrophages extend long pseudopodia, while others make intimate contact with lymphocytes, suggesting their high level of activity.
Similar content being viewed by others
References
Abrahams C (1964) The ultrastructure of the interstitial tissue of the renal papilla of the rat. S Afr J Med Sci 29:17–20
Abrahams, Pirani CL (1966) The renal papilla of the rat: Electronmicroscopic and histochemical studies. S Afr J Med Sci 31:107–117
Bohman S-O (1974) The ultrastructure of the rat renal medulla as observed after improved fixation methods. J Ultrastruct Res 47:329–360
Bojesen I (1974) Quantitative and qualitative analyses of isolated lipid droplets from interstitial cells in renal papillae from various species. Lipids 9:835–843
Brown CA, Zusman RM, Haber E (1980) Identification of an angiotensin receptor in rabbit renomedullary interstitial cells in tissue culture. Circ Res 46:802–807
Bulger RE, Nagle RB (1973) Ultrastructure of the interstitium in the rabbit kidney. Am J Anat 136:183–204
Bulger RE, Trump BF (1966) Fine structure of the rat renal papilla. Am J Anat 118:685–722
Dieterich HJ (1978) Die Struktur der Blutgefäße in der Rattenniere. In: Bargmann W, Doerr W (eds) Normale und pathologische Anatomie, vol 35. Thieme, Stuttgart, pp 1–108
Falchuk KH, Berliner RW (1971) Hydrostatic pressures, in peritubular capillaries and tubules in the rat kidney. Am J Physiol 220:1422–1426
Fourman J, Moffat DB (1971) The blood vessels of the kidney. Blackwell Scientific Publications, Oxford Edinburgh, pp 90–114
Fujita T (1978) Microarchitecture of reticular tissues. Reevaluation of the RES by scanning electron microscopy. Recent Adv RES Res 18:1–19
Fujiwara T, Uehara Y (1984) The cytoarachitecture of the wall and the innervation pattern of the microvessels in the rat mammary gland: A scanning electron microscopic observation. Am J Anat 170:39–54
Gottschalk CW, Mylle M (1957) Micropuncture study of pressures in proximal and distal tubules and peritubular capillaries of the rat kidney during osmotic diuresis. Am J Physiol 189:323–328
Kaneda K, Dan C, Wake K (1983) Pit cells as natural killer cells. Biomed Res 4:567–576
Kessel RG, Kardon RH (1979) Tissue and organs; a text-atlas of scanning electron microscopy. Freeman and Company, San Francisco
Kirkman H (1943) The number and distribution of macrophages and fibroblasts in kidneys of albino rats with emphasis on twenty-five day males. Am J Anat 73:451–482
Kriz W (1981) Structural organization of the renal medulla: comparative and functional aspects. Am J Physiol 241:R3-R16
Latta H, Maunsbach AB, Osvaldo L (1967) The fine structure of renal tubules in cortex and medulla. In: Dalton AJ, Haguenau F (eds) Ultrastructure of the kidney. Academic Press, New York, pp 1–56
McGiff JC, Crowshaw K, Terrango NA, Lonigro AJ (1970) Release of a prostaglandin-like structure into renal venous blood in response to angiotensin II. Circ Res 26, 27 [Suppl I]:121–130
Möllendorff W von (1930) Harn-und Geschlechtsapparat. Hand-buch der mikroskopischen Anatomie des Menschen, VII/1. Springer, Berlin Heidelberg New York
Moffat DB (1967) The fine substance of the blood vessels of the renal medulla with particular reference to the control of the medullary circulation. J Ultrastruct Res 19:532–545
Muehrcke RC, Mandal AK, Volini FI (1970) A pathophysiological review of the renal medullary interstitial cells and their relationship to hypertension. Circ Res 26, 27 [Suppl I]:109–119
Murakami T (1974) A revised tannin-osmium method for noncoated scanning electron microscope specimens. Arch Histol Jpn 36:189–193
Novikoff AB (1960) The rat kidney: cytochemical and electron microscopic studies. In: Quinn EL, Kass EH (eds) Biology of pyelonephritis. Little Brown and Company, Boston, Mass, pp 113–144
Osvaldo L, Latta H (1966) Interstitial cells of the renal medulla. J Ultrastruct Res 15:589–613
Owen RL, Bhalla DK (1983) Lympho-epithelial organs and lymph nodes. In: Hodges GM, Carr KE (eds) Biomedical research applications of scanning electron microscopy, vol 3. Academic Press, London New York, pp 79–169
Renaut J, Dubreuil G (1907) Note sur l'histologie, la cytologie des tubes de Bellini et le tissu conjonctif de la pyramide du rein. C R Assoc Anat 9:94–103
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–211
Romen W, Thoenes W (1970) Histocytäre und fibrocytäre Eigenschaften der interstitiellen Zellen der Nierenrinde. Virchows Arch [B] 5:365–375
Roszkiewicz A, Roszkiewicz J, Zawistowski S (1982) The effect of indomethacin on the ultrastructure of renomedullary interstitial cells of the rat. Gegenbaurs Morphol Jahrb 128:702–711
Rühle G (1897) Uber die Membrana propria der Harnkanälchen und ihre Beziehung zu dem interstitiellen Gewebe der Niere. Arch Anat Entwicklungsgesch 21:153–170
Schiller A, Taugner R (1979) Junctions between interstitial cells of the renal medulla: a freeze-fracture study. Cell Tissue Res 203:231–240
Schwartz MM, Karnovsky MJ, Venkatachalam MA (1976) Ultrastructural differences between rat inner medullary descending and ascending vasa recta. Lab Invest 35:161–170
Shimada T, Nakamura M, Inoue Y (1981) Removal of extracellular materials by HCl-tween treatment. Arch Histol Jpn 44:189–192
Sternberg WH, Farber E, Dunlap CE (1956) Histochemical localization of specific oxidative enzymes: II. Localization of diphosphopyridine nucleotide and triphospyridine nucleotide diaphorases and the succindehydrogenase system in the kidney. J Histochem Cytochem 4:266–283
Takahashi-Iwanaga H (1989) The three-dimensional structure of renal tubule cells. In: Motta PM (ed) Cells and tissues: a threedimensional approach by modern techniques in microscopy. Liss, New York, pp 203–212
Takahashi-Iwanaga H, Fujita T (1986) Application of an NaOH maceration method to a scanning electron microscopic observation of Ito cells in the rat liver. Arch Histol Jpn 49:349–357
Takahashi-Iwanaga H, Iwata Y, Adachi K, Fujita T (1989) The histotopography and ultrastructure of the thin limb of the Henle's loop: a scanning electron microscopic study of the rat kidney. Arch Histol Cytol 52:395–405
Vimtrup B, Schmidt-Nielsen B (1952) The histology of the kidney of kangaroo rats. Anat Rec 114:515–528
Zimmermann KW (1923) Der feinere Bau der Blutcapillaren. Z Anat Entwicklungsgesch 68:29–109
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi-Iwanaga, H. The three-dimensional cytoarchitecture of the interstitial tissue in the rat kidney. Cell Tissue Res 264, 269–281 (1991). https://doi.org/10.1007/BF00313964
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00313964