Abstract
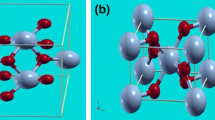
The O2− ion is a useful concept in ionic solids. Its electron density and large polarizability is well described by the Watson sphere model. The large variation in the electronic structure of oxides is illustrated by discussing the ionic MgO, the partly ‘covalent’ Cu2O, and TiO and NbO which have defects with respect to the ideal NaCl structure. The variation in physical properties is shown for the oxides in the rutile structure ranging from the insulating TiO2, the metallic RuO2 to the ferromagnetic CrO2. Electron densities, total energies and densities of states are used to study these materials.
Similar content being viewed by others
References
Blaha P, Schwarz K (1983) Electron densities and chemical bonding in TiC, TiN, and TiO derived from energy band calculations. Int J Quantum Chem 23:1535–1552
Bussmann A, Bilz H, Roespiess R, Schwarz K (1980) Oxygen polarizability in ferroelectric phase transitions. Ferroelectrics 25:343–346
Henrich VE (1985) The surfaces of metal oxides. Rep Progr Phys 48:1481–1541
Huisman L, Carlsson AE, Gelatt CD Jr, Ehrenreich H (1980) Mechanisms for energetic-vacancy stabilization: TiO and TiC. Phys Rev B 22:991–1006
Marksteiner P, Blaha P, Schwarz K (1986) Electronic structure and binding mechanism of Cu2O. Z Physik B — Condensed Matter 64:119–127
Mattheiss LF (1976) Electronic structure of RuO2, OsO2, and IrO2. Phys Rev B 13:2433–2450
Nagel S (1985) Nuclear quadrupole interaction and chemical bonding. MSXα Cluster calculation for Cu2O. J Phys Chem Solids 46:743–756
Neckel A (1983) Recent investigations on the electronic structure of the fourth and fifth group transition metal monocarbides, mononitrides, and monoxides, Int J Quantum Chem 23:1317–1353
Redinger J, Schwarz AK (1981) Electronic charge distribution of the polarizable O2− ion in MgO and CaO in contrast to the F− ion in NaF. Z Physik B — Condensed Matter 40:269–276
Schwarz K (1984) Band structure and non-stoichiometry of metallic oxides. In: Dominguez-Rodriguez A, Castaing J, Marquez R (eds) Basic Properties of Binary Oxides. Servicio de Publicationes de la Universidad de Sevilla (Serie Ciencias) pp 43–56
Schwarz K (1986) CrO2 predicted as half-metallic ferromagnet. J Phys F: Metal Phys 16:L211-L215
Schwarz K, Blaha P (1984) Electron densities in solid compounds. In: Dahl JP, Avery J (eds) Local Density Approximation in Quantum Chemistry and Solids. Plenum Press, New York, pp 605–616
Schwarz K, Schulz H (1978) X-ray scattering factors for O2− and N3−. Acta Crystallogr A34:994–999
Terakura K, Williams AR, Oguchi T, Kübler J (1984) Transitionmetal monoxides: Band or Mott insulators. Phys Rev Lett 52:1830–1833
Watson RE (1958) Analytic Hartree-Fock solutions for O2−. Phys Rev 111:1108–1110
Williams AR, Kübler J, Gelatt CD Jr (1979) Cohesive properties of metallic compounds: augmented-spherical-wave calculations. Phys Rev B19:6094–6118
Wimmer E, Schwarz K, Podoucky, Herzig P, Neckel A (1982) The effect of vacancies on the electronic structure of NbO. J Phys Chem Solids 43:439–447
Author information
Authors and Affiliations
Additional information
Paper from Conference on Quantum Theory and Experiment, July 1986
Rights and permissions
About this article
Cite this article
Schwarz, K. Band theoretical studies of the electronic structure of oxides. Phys Chem Minerals 14, 315–319 (1987). https://doi.org/10.1007/BF00309803
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00309803