Summary
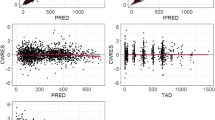
Pharmacokinetic studies in 11 patients with multiple myeloma were undertaken on the first and last days of one course of chemotherapy. The drug was administered PO in single doses of 6–14 mg daily. Melphalan concentrations were determined by high-performance liquid chromatography. The interpatient variability of pharmacokinetic parameters noted by other authors was observed. Regression analysis showed a significant positive correlation between the elimination rate constant for melphalan and renal function (P=0.003). The form of the line which describes the overall elimination rate constant for melphalan is given by the equation: Kel=5.67×10-3+[4.90×10-5xGFR]. There was also a significant negative correlation between renal function and the area under the plasma melphalan concentration/time curve (P=0.006). In vitro stability studies of melphalan in plasma at 37°C and pharmacokinetic data suggest that hydrolysis and renal clearance are the major mechanisms of melphalan elimination. This work shows quantitatively the relationship between renal function and drug elimination and how the data may be used in predicting melphalan half-life from creatinine clearance.
Similar content being viewed by others
References
Adair CG, Burns DT, Crockard AD, Desai ZR, Harriott M (1984) Modified extraction and chromatography for the measurement of plasma melphalan by ion pair high performance liquid chromatography. J Chromatogr [Biomed Appl] 336: 429
Alberts DS, Chang SY, Chen H-SY, Evans TL, Moon TE (1979) Oral melphalan kinetics. Clin Pharmacol Ther 26: 737
Alberts DS, Chang SY, Chen H-SY, Larcom BJ, Evans TL (1980) Comparative pharmacokinetics of chlorambucil and melphalan in man. Recent Results Cancer Res 74: 124
Alberts DS, Chen H-SY, Berg D, Mason DL (1981) Effects of renal dysfunction in dogs on the disposition and marrow toxicity of melphalan. Br J Cancer 43:330
Bosanquet AG, Gilby ED (1982) Pharmacokinetics of oral and intravenous melphalan during routine treatment of multiple myeloma. Eur J Clin Oncol 18: 355
Bosanqeut AG, Gilby ED (1984) Comparison of the fed and fasting states on the absorption of melphalan in multiple myeloma. Cancer Chemother Pharmacol 12: 183
Cockcroft DW, Gault MH (1976) Prediction of creatinine clearance from serum creatinine. Nephron 16: 31
Cornwell GG, Pajak TF, McIntyre OR, Kochwa S, Dosik D (1982) Influence of renal failure on the myelosuppressive effects of melphalan: Cancer and Leukaemic Group B experience. Cancer Treat Rep 66: 475
Evans TL, Chang SY, Alberts DS, Sipes G, Brendel K (1982) In vitro degradation of L-phenylalanine mustard (L-PAM). Cancer Chemother Pharmacol 8: 175
Furner RL, Brown RK (1980) Melphalan: The first 25 years. Cancer Treat Rep 64: 559
Gobbi M, Cavo M, Savelli G, Baccurani M, Tura S (1980) Prognostic factors and survival in multiple myeloma. Haematologica (Pavia) 65: 437
Oken MM (1984) Multiple myeloma. Med Clin North Am 68: 757
Taha AK, Ahmed RE, Gray H, Roberts CI, Rogers HS (1982) Plasma melphalan and prednisolone concentrations during oral therapy for multiple myeloma. Cancer Chemother Pharmacol 9: 57
Author information
Authors and Affiliations
Additional information
The work described in this paper was supported by the Northern Ireland Leukaemia Research Fund
Rights and permissions
About this article
Cite this article
Adair, C.G., Bridges, J.M. & Desai, Z.R. Renal function in the elimination of oral melphalan in patients with multiple myeloma. Cancer Chemother. Pharmacol. 17, 185–188 (1986). https://doi.org/10.1007/BF00306752
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00306752