Abstract
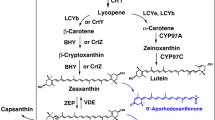
Erwinia herbicola is a nonphotosynthetic bacterium that is yellow pigmented due to the presence of carotenoids. When the Erwinia carotenoid biosynthetic genes are expressed in Escherichia coli, this bacterium also displays a yellow phenotype. The DNA sequence of the plasmid pPL376, carrying the entire Erwinia carotenoid gene cluster, has been found to contain 12 open reading frames (ORFs). Six of the ORFs have been identified as carotenoid biosynthesis genes that code for all the enzymes required for conversion of farnesyl pyrophosphate (FPP) to zeaxanthin diglucoside via geranylgeranyl pyrophosphate, phytoene, lycopene, β-carotene, and zeaxanthin. These enzymatic steps were assigned after disruption of each ORF by a specific mutation and analysis of the accumulated intermediates. Carotenoid intermediates were identified by the absorption spectra of the colored components and by high pressure liquid chromatographic analysis. The six carotenoid genes are arranged in at least two operons. The gene coding for β-carotene hydroxylase is transcribed in the opposite direction from that of the other carotenoid genes and overlaps with the gene for phytoene synthase.
Similar content being viewed by others
References
Altschul SF, Warren G, Miller W, Myers E, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Armstrong GA, Alberti M, Leach F, Hearst JE (1989) Nucleotide sequence, organization, and nature of the protein products of the carotenoid biosynthesis gene cluster of Rhodobacter capsulatus. Mol Gen Genet 216:254–268
Armstrong GA, Alberti M, Hearst JE (1990) Conserved enzymes mediate the early reactions of carotenoid biosynthesis in nonphotosynthetic and photosynthetic prokaryotes. Proc Natl Acad Sci USA 87:9975–9979
Armstrong GA, Hundle BS, Hearst JE (1993) Evolutionary conservation and structural similarities of carotenoid biosynthesis gene products from photosynthetic and nonphotosynthetic organisms. Methods Enzymol 214:297–311
Barany F (1985) Single-stranded hexameric linkers: a system for in-phase insertion mutagenisis and protein engineering. Gene 37:111–123
Fraser PD, Misawa N, Linden H, Yamano S, Kobayashi K, Sandmann G (1992) Expression in Escherichia coli, purification, and reactivation of the recombinant Erwinia uredovora phytoene desaturase. J Biol Chem 267:19891–19895
Hundle BS, Beyer P, Kleinig H, Englert G, Hearst JE (1991) Carotenoids of Erwinia herbicola and an Escherichia coli HB101 strain carrying the Erwinia herbicola carotenoid gene cluster. Photochem Photobiol 54:89–93
Hundle BS, O'Brien DA, Alberti M, Beyer P, Hearst JE (1992). Functional expression of zeaxanthin glucosyltransferase from Erwinia herbicola and a proposed uridine diphosphate binding site. Proc Natl Acad Sci USA 89:9321–9325
Hundle BS, O'Brien DA, Beyer P, Kleinig H, Hearst JE (1993) In vitro expression and activity of lycopene cyclase and β-carotene hydoxylase from Erwinia herbicola. FEBS Lett 315:329–334
Hunter CN, Hundle BS, Hearst JE, Lang HP, Gardiner AT, Takaichi S, Cogdell RJ (1994) Introduction of new carotenoids into the bacterial photosynthetic apparatus by splicing the genes for carotenoid biosynthetic pathways of Erwinia herbicola and Rhodobacter sphaeroides. J Bacteriol 176:3692–3697
Lee L-Y, Liu S-T (1991) Characterization of the yellow-pigment genes of Erwinia herbicola. Mol Microbiol 5:217–224
Liu S-T (1993) Carotenoid-biosynthesis genes as a genetic marker for the purpose of gene cloning. Biochem Biophys Res Commun 195:259–263
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Marrs B (1981) Mobilization of the genes for photosynthesis from Rhodobacter capsulata by a promiscuous plasmid. J Bacteriol 146:1003–1012
Math SK, Hearst JE, Poulter DC (1992) The crtE gene in Erwinia herbicola encodes geranylgeranyl diphosphate synthase. Proc Natl Acad Sci USA 89:6761–6764
McClure WR (1985) Mechanism and control of transcription initiation in prokaryotes. Annu Rev Biochem 54:171–204
Misawa N, Nakagawa M, Kobayashi K, Yamano S, Izawa Y, Nakamura K, Harashima K (1990) Elucidation of the Erwinia uredovora carotenoid biosynthetic pathway by functional analysis of gene products expressed in Escherichia coli. J Bacteriol 172:6704–6712
Nagy M, Lacroute F, Dominique T (1992) Divergent evolution of pyrimidine biosynthesis between anaerobic and aerobic yeasts. Proc Natl Acad Sci USA 89:8966–8970
Ohnuma S, Suzuki M, Nishino T (1994) Archaebacterial etherlinked lipid biosynthetic gene. Expression cloning, sequencing, and characterization of geranylgeranyl-diphosphate synthase. J Biol Chem 269:14792–14797
Perry KC, Simonitch TA, Harrison-Lavoie KJ, Liu ST (1986) Cloning and regulation of Erwinia herbicola pigment genes. J Bacteriol 168:607–612
Sandmann G, Misawa N (1991) New functional assignment of the carotenogenic genes crtB and crtE with constructs of these genes from Erwinia species. FEMS Lett 90:253–258
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Straub O (1987) In: Pfander H, Rychener M. Schwabe R (eds) Key to carotenoids. BirkMuser Verlag; pp 32–90
Tuveson RW, Larson RA, Kagan J (1988) Role of cloned carotenoid genes expressed in Escherichia coli in protecting against inactivation by near-UV light and specific phototoxic molecules. J Bacteriol 170:4675–4680
Weidemann M, Misawa N, Sandmann G (1993) Purification and enzymatic characterization of the geranylgeranyl pyrophosphate synthase from Erwinia uredovora after expression in Escherichia coli. Arch Biochem Biophys 306:152–157
Wierenga RK, Terpstra P, Hol WGJ (1986) Prediction of the occurrence of the ADP-binding βαβ-fold in proteins, using an amino acid sequence fingerprint. J Mol Biol 187:101–107
Author information
Authors and Affiliations
Additional information
Communicated by A. Kondorosi
Rights and permissions
About this article
Cite this article
Hundle, B., Alberti, M., Nievelstein, V. et al. Functional assignment of Erwinia herbicola Eho10 carotenoid genes expressed in Escherichia coli . Molec. Gen. Genet. 245, 406–416 (1994). https://doi.org/10.1007/BF00302252
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00302252