Summary
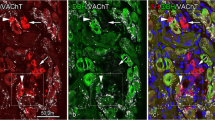
Combining glyoxylic acid and pontamine sky blue fluorescence methods allows simultaneous visualization of catecholamine fibers and the intra-hippocampal vascular bed. The normal noradrenergic innervation of the hippocampal formation is not closely associated with the vasculature in any region. However, following lesions of the septo-hippocampal fibers, norepinephrine-containing sympathetic fibers course along the penetrating arterioles into the parenchyma of regio inferior and the area dentata. Within the respective cell layers, the anomalous sympathetic axons collateralize freely, thus suggesting a dual relationship with the vasculature and with the neuropil of selective hippocampal zones.
Similar content being viewed by others
References
Banker GA (1980) Trophic interactions between astroglial cells and hippocampal neurons in culture. Science 209:809–810
Bloom FE, Battenberg ELF (1976) A rapid, simple and sensitive method for the demonstration of central catecholamine-containing neurons and axons by glyoxylic acid-induced fluorescence. II. A detailed description of methology. J Histochem Cytochem 24:561–571
Coyle P (1978) Spatial features of the rat hippocampal vascular system Exp Neurol 58:549–561
Crutcher KA, Brothers L, Davis JN (1979) Sprouting of sympathetic nerves in the absence of efferent input. Exp Neurol 66:778–783
Koelle GB (1954) The histochemical localization of cholinesterases in the central neurvous system of the rat J Comp Neurol 100:211–235
Konig J, Klippel R (1967) The rat brain. Krieger Publ Co New York
Loy R, Milner TA (1980) Sexual dimorphism in extent of axonal sprouting in hippocampus. Science 208:1282–1284
Loy R, Moore RY (1977) Anomalous innervation of the hippocampal formation by peripheral sympathetic axons following mechanical injury. Exp Neurol 57:645–650
Loy R, Koziell DA, Lindsey JD, Moore RY (1980a) Noradrenergic innervation of the adult rat hippocampal formation. J Comp Neurol 189:699–710
Loy R, Milner TA, Moore RY (1980b) Sprouting of sympathetic axons in the hippocampal formation: Conditions necessary to elicit ingrowth. Exp Neurol 67:399–411
McGinty JF, Koda LY, Bloom FE (1979) A novel fluorescent marker of CNS vasculature used in combination with monoamine histofluorescence. Neurosci Abstr 5:344
Milner TA, Loy R (1980a) A delayed sprouting response to partial hippocampal deafferentation. Time course of sympathetic ingrowth following fimbrial lesions Brain Res 197:391–399
Milner TA, Loy R (1980b) Interaction of age and sex in sympathetic axon ingrowth into the hippocampus following septal afferent damage Anat Embryol 161:159–168
Scheff SW, Benardo LA, Cotman CW (1978a) Lesion-induced vascular changes in the dentate gyrus following removal of the entorhinal cortex in adult rats Exp Neurol 62:815–820
Scheff SW, Benardo L, Cotman CW (1978b) Decrease in adrenergic sprouting in senescent rat Science 202:775–778
Stenevi U, Björklund A (1978) A pitfall in brain lesion studies: Growth of vascular sympathetic axons into the hippocampus after fimbrial lesions Neurosci Lett 1:219–224
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McGinty, J.F., Milner, T.A. & Loy, R. Association of sympathetic axons in denervated hippocampus to intracerebral vasculature. Anat Embryol 164, 95–100 (1982). https://doi.org/10.1007/BF00301882
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00301882