Summary
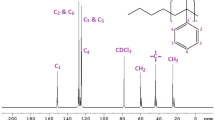
Polystyrene macromonomers with terminal 1,1-diphenylethylene functionality were prepared by the reaction of one equivalent of poly(styryl)lithium with 1,4-bis (l-phenylethenyl)benzene (PDDPE). The macromonomer functionalities were determined by 1H NMR [σ(vinyl CH2)=5.4 ppm] and UV spectroscopy (λmax=260 nm). The stoichiometric linking reaction of poly(styryl)lithium (Mn=15.3x103 g/mol) with an ω-1,1-diphenylethylene-terminated polystyrene macromonomer (Mn=5.4x103 g/mol) followed by addition of styrene monomer has been used to prepare a hetero three-armed, star-branched polymer with Mn=5.8x104 g/mol (5,400-15,300-37,300). The g′ value ([ν]b/[ν]l) was equal to 0.92.
Similar content being viewed by others
References
Roovers J (1985) Branched polymers. in: Kroschwitz JI (ed) Encyclopedia of Polymer Science and Engineering. Wiley-Interscience, New York (vol 2 pp 478)
Bywater S (1979) Adv. Polym. Sci. 30:89
Bauer BJ, Fetters LJ (1978) Rubber Chem. Tech. 51:406
Rempp P, Herz JE (1989) Model polymers. in: Kroschwitz JI (ed) Encyclopedia of Polymer Science and Engineering. Wiley-Interscience, New York (supplement vol pp 493)
Rempp P, Franta E, Herz J-E (1988) Adv. Polym. Sci. 86:145
Morton M (1983) Anionic Polymerization: Principles and Practice. Academic Press, New York
Pennisi RW, Fetters LJ (1988) Macromolecules 21:1094
Khasat N, Pennisi RW, Hadjichristidis N, Fetters LJ (1988) Macromolecules 21:1100
Iatrou H, Hadjichristidis N (1992) Macromolecules 25:4649
Mays J (1990) Polym. Bull. 23:247
Tsitsilianis C, Chaumont P, Rempp P (1990) Makromol. Chem. 191:2319
Tsitsilianis C, Graff S, Rempp P (1991) Eur. Polym. J. 27:243
Quirk RP, Lee B, Schock LE (1992) Makromol. Chem., Macromol. Symp. 53:201.
Quirk RP, Ignatz-Hoover F (1987) Living coupling agents. in: Hogen-Esch TE, Smid I (ed) Recent Advances in Anionic Polymerization. Elsevier, New York (pp 393)
Quirk RP, Chen W-C (1982) Makromol. Chem. 183:2071
Quirk RP, Cheng PL (1986) Macromolecules 19:1291
Gilman H, Cartledge, FK (1964) J. Organomet. Chem. 2:447
Höcker H, Lattermann G (1972) Makromol. Chem. 158:191
Shriver DF, Drezdzon MA (1986) The Manipulation of Air-Sensitive Compounds. 2nd ed, Wiley-Interscience, New York
Morton M, Fetters LJ (1975) Rubber Chem. Tech. 48:359
Brown TL, Dickerhoof DW, Bafus DA, Morgan GL (1962) Rev. Sci. Instrum. 33:49
Grubisic Z, Rempp P, Benoit H (1967) J. Polym. Sci., Polym. Lett. 5:753
Boni KA, Sliemers FA, Stickney PB (1968) J. Polym. Sci. A-2, 6:1567
Tung LH, Runyan RJ (1973) J. Appl. Polym. Sci. 17:1589
Tung LH (1979) J. Appl. Polym. Sci. 24:953
Quirk RP, Ma J-J (1991) Polym. Int. 24:197
Leitz E, Hocker H (1983) Makromol. Chem. 184:1893
Broske AD, Huang TL, Allen RD, Hoover JM, McGrath JE (1987) Investigations of hydrocarbon soluble difunctional organolithium initiators based upon 1,3-bis(phenyl ethenyl) benzene in: Hogen-Esch TE, Smid I (ed) Recent Advances in Anionic Polymerization. Elsevier, New York (pp 363)
Quirk RP, Ma J-J (1988) Polym. Prepr., Am. Chem. Soc., Div. Polym. Chem. 29(2):10
Ma J-J (1991) Ph.D. Thesis, University of Akron
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quirk, R.P., Yoo, T. Anionic synthesis of ω-1,1-diphenylethylene-terminated polystyrene macromonomers. Rational synthesis of ABC hetero three-armed-star-branched polymers. Polymer Bulletin 31, 29–36 (1993). https://doi.org/10.1007/BF00298760
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00298760