Summary
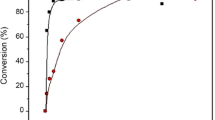
Zn lactate, 3H2O and dehydrated Zn lactate were used as initiator to polymerize DL-lactide under various conditions. It is shown that the latter does polymerize DL-lactide up to high molecular weight at a slower rate than Sn octoate. Zn lactate being formed when lactic acid is allowed to react with Zn metal, it is believed that the active species in Zn metal initiation is Zn lactate. Comparison was made of the two poly(DL-lactide) initiated with Zn metal and Sn octoate respectively and selected to have almost similar characteristics. It is shown that degradation characteristics of the two polymers are different, especially water uptake and rate of heterogeneous degradation. It is further shown that residual Sn concentrated within the matrix remnants whereas the content in Zn remained constant according to degradation controlled release.
Similar content being viewed by others
References
Kleine J. and H. (1959) Makromol. Chem., 30: 23
Kricheldorf H.R., Jonte J.M. and Berl M. (1995) Makromol. Chem.: Suppl. 12: 25
Chabot F., Vert M., Chapelle S. and Granger P. (1983) Polymer, 24: 53
Schwach G., PhD thesis, University Montpellier 1, France, Feb. 22, 1996
Bero M., Kasperczyk J. and Jedlinski Z. (1990) Makromol. Chem., 191: 2287
Li S.M. and Vert M. (1995) Degradable Polymers: Principles and Applications, Scott G. and Gilead D. Eds, Chapman & Hall, London, p. 43
Li S.M., Garreau H. and Vert M. (1990) J. Mater. Sci.: Mater. in Med., 1: 23
Espartero-Sanchez J.L., Rashkov I., Li S.M., Manolova N. and Vert M. (1996) Macromolecules, 29, 3535
Schwach G, Engel R., Coudane J. and Vert M. (1994) Polym. Bull., 32, 617
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwach, G., Coudane, J., Engel, R. et al. Zn lactate as initiator of dl-lactide ring opening polymerization and comparison with Sn octoate. Polymer Bulletin 37, 771–776 (1996). https://doi.org/10.1007/BF00295776
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00295776