Summary
Xylose, glucose and xylose/glucose mixtures were fermented with Candida tropicalis ATCC 32113 under aerobic, oxygen limited and anaerobic conditions. Ethanol yields were highest under oxygen limited conditions with xylose and xylose/glucose. Anaerobic conditions were best for glucose fermentations.
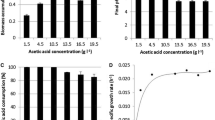
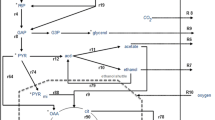
The effect of four metabolic inhibitors (azide, carbonyl cyanide m-chlorophenyl hydrazone (CCCP), oligomycin A and valinomycin-K+) were then studied under oxygen limited conditions. Only azide had a significant influence on ethanol production. At 2¢10-4 M concentrations, ethanol yield increased up to two times and xylitol levels were repressed by 90% for xylose and glucose/xylose fermentations. 4.2×10-3 M azide gave highest ethanol yields in glucose fermentations. At this concentration of azide, however, cell growth was inhibited, which seemed to prevent ethanol production in xylose fermentations. The effect of azide is discussed in terms of “fine-tuning” the respiratory activity necessary for metabolism.
Similar content being viewed by others
References
Ainsworth GC, Sussman AS (1968) The fungi, vol 3. Academic Press, New York
Cooper CM, Fernstrom GA, Miller SA (1944) Performance of agitated gas-liquid contactors. Ind Eng Chem 36:506–509
Debus D, Methner H, Schulze D, Dellweg H (1983) Fermentation of xylose with the yeast Pachysolen tannophilus. Eur J Appl Microbiol Biotechnol 17:287–291
Gong CS, Mc Craken LD, Tsao GT (1981) Direct fermentation of d-xylose to ethanol by a xylose-fermenting yeast mutant Candida SP XF 217. Biotechnol Lett 3:245–250
Hahn-Hägerdal B (1983) Fermentation of enzymatically hydrolyzed salix using yeast and bacteria. Workshop über Verwertungsmöglichkeiten von Pentosen, 19/20 Jan 1983, Technische Universität Wien
Hahn-Hägerdal B, Mattiasson B (1982) Azide sterilization of fermentation media. Eur J Appl Microbiol Biotechnol 14:140–143
Heytler PG (1979) Uncouplers of oxidative phosphorylation. In: Eleischer S, Packer L (eds) Methods in enzymology, vol LV. Academic Press, New York, pp 462–472
Horecker BL (1962) Pentose metabolism in bacteria. John Wiley ans Sons Inc, New York
Jeffries TW (1981) Conversion of xylose to ethanol under aerobic conditions by Candida tropicalis. Biotechnol Lett 3:213–218
Kaplan NO, Girotti MM (1957) Enzymatic determination of ethanol. In: Colewick SP, Kaplan NO (eds) Methods in enzymology III. Academic Press, New York, p 253
Margaritis A, Bajpaj P (1982). Direct fermentation of d-xylose to ethanol by Kluyveromyces marxianus strains. Appl Environ Microbiol 44:1039–1041
Reed PW (1979) Ionophores. In: Fleischer S, Packer L (eds) Methods in enzymology, vol LV. Academic Press, New York, pp 435–454
Schneider H, Wang PY, Chan YK, Maleszka R (1981) Conversion of d-xylose into ethanol by the yeast Pachysolen tannophilus. Biotechnol Lett 3:89–92
Slater EC (1967) Application of inhibitors and uncouplers for a study of oxidative phosphorylation. In: Pullman ME (ed) Methods in enzymolgoy, vol X. Academic Press, New York, pp 48–57
Slinger PL, Bothast, RJ, van Cauwenberge JE, Kurtzman CP (1982) Conversion of d-xylose to ethanol by the yeast Pachysolen tannophilus. Biotechnol Bioeng 24:371–384
Smiley KL, Bolen PL (1982) Demonstration of d-xylose reductase and d-xylitol dehydrogenase in Pachysolen tannophilus. Biotechnol Lett 4:607–610
Sols A, Gancedo C, DelaFuente G (1971) Energy yielding metabolism in yeasts. In: Rose AH, Harrison JS (eds) The yeasts, vol 2. Academic Press, New York pp 271–307
Stickland LH (1956) The pasteur effect in normal yeast and its inhibition by various agents. Biochem J 64:503–515
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lohmeier-Vogel, E., Hahn-Hägerdal, B. The utilization of metabolic inhibitors for shifting product formation from xylitol to ethanol in pentose fermentations using Candida tropicalis . Appl Microbiol Biotechnol 21, 167–172 (1985). https://doi.org/10.1007/BF00295113
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00295113