Summary
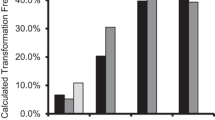
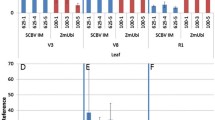
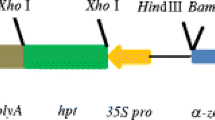
It has been previously reported that the 5′ region of the rice actin 1 gene (Act1) promoted high-level expression of a β-glucuronidase reporter gene (Gus) in transformed rice cells. In this paper we describe the construction of Act1-based expression vectors for use in monocot transformation. As part of the development of these vectors, we have evaluated the influence of the Act1 first intron, the Act1-Gus junction-encoded N-terminal amino acids, and the sequence context surrounding the Act1 and Gus translation initiation site on Act1-Gus gene expression in rice and maize cells. We have found that addition of Act1 intron 1 to the transcription unit of a Gus reporter gene under control of the cauli-flower mosaic virus (CaMV) 35S promoter stimulated GUS activity more than 10-fold in transformed rice cells. Optimization of the sequence context around the Gus translation initiation site resulted in a 4-fold stimulation of Gus expression in transformed rice cells. By utilizing both the Act1 intron 1 and optimized Gus translation initiation site, a 40-fold stimulation in Gus expression from the CaMV 35S promoter has been achieved in transformed rice cells; very similar results were obtained in transformed maize cells. Taken together these results suggest that the Act1-based expression vectors described here should promote the expression of foreign genes in most, if not all, transformed monocot cells to levels that have not previoulsy been attainable with alternative expression vectors.
Similar content being viewed by others
References
Bachmair A, Finley D, Varshavsky A (1986) In vivo half-life of a protein is a function of its amino-terminal residue. Science 234:179–186
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye-binding. Anal Biochem 72:248–254
Callis J, Fromm M Walbot V (1987) Introns increase gene expression in cultured maize cells. Genes Dev 1:1183–1200
Dennis ES, Gerlach WL, Pryor AJ, Bernetzen JL, Inglis A, Llewellyn D, Sachs MM, Ferl RJ, Peacock WJ (1984) Molecular analysis of the alcohol dehydrogenase (Adh1) gene of maize. Nucleic Acids Res 12:3983–4000
Ellis JG, Llewellyn DJ, Dennis ES, Peacock WJ (1987) Maize Adh1 promoter sequences control anaerobic regulation: addition of upstream promoter elements from constitutive genes is necessary for expression in tobacco. EMBO J 6:11–16
Fromm ME, Morrish F, Armstrong C, Williams R, Thomas J, Klein TM (1990) Inheritance and expression of chimeric genes in the progeny of transgenic maize plants. Bio/Technology 8:833–839
Gonda DK, Bachmair A, Wunning I, Tobias JW, Lane WS, Varshavsky A (1989) Universality and structure of the N-end rule. J Biol Chem 264:16700–16712
Gordon-Kamm WJ, Spencer TM, Mangano ML, Adams TR, Dames RJ, Start WG, O'Brien V, Chambers SA, Adams WR, Willetts NG, Rice TB, Mackey CJ, Krueger RW, Kausch AP, Lemaux PG (1990) Transformation of maize cells and regeneration of fertile transgenic plants. Plant Cell 2:603–618
Guilley H, Dudley RK, Jonard G, Balàzs E, Richards KE (1982) Transcription of cauliflower mosaic virus DNA: Detection of promoter sequences, and characterization of transcripts. Cell 30:763–773
Jefferson RA (1987) Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol Biol Rep 5:387–405
Jenes B, Pauk J (1989) Plant regeneration from protoplast derived calli in rice (Oryza sativa) using dicamba. Plant Sci 63:187–197
Joshi CP (1987) An inspection of the domain between putative TATA box and translation start site in 79 plant genes. Nucleic Acids Res 15:6643–6653
Keith B, Chua N-H (1986) Monocot and dicot pre-mRNAs are processed with different efficiencies in transgenic tobacco. EMBO J 5:2419–2425
Klein TM, Gradzeil T, Fromm ME, Sanford JC (1988) Factors influencing delivery into Zea mays cells by high-velocity microprojectiles. Bio/Technology 6:559–653
Kozak M (1984) Compilation and analysis of sequences upstream from the translation start site in eukaryotic mRNAs. Nucleic Acids Res 12:857–872
Kozak M (1986) Point mutations define a sequence flanking the AUG initiator codon that modulates translation by eukaryotic ribosomes. Cell 44:283–292
Kozak M (1987) At least six nucleotides preceding the AUG initiator codon enhance translation in mammalian cells. J Mol Biol 196:947–950
Kunkel TA (1985) Rapid and efficient site-specific mutagenesis without phenotypic selection. Proc Natl Acad Sci USA 82:488–492
Kyozuka J, Izawa M, Nakajima M, Shimamoto K (1990) Effect of the promoter and the first intron of maize Adh1 on foreign gene expression in rice. Maydica 35:353–357
Kyozuka J, Fujimoto H, Izawa T, Shimamoto K (1991) Anaerobic induction and tissue specific expression of maize Adh1 promoter in transgenic rice plants and their progeny. Mol Gen Genet (in press)
Liu C-C, Simonsen CC, Levinson AD (1984) Initiation of translation at internal AUG codons in mammalian cells. Nature 309:82–85
Luehrsen KR, Walbot V (1991) Intron enhancement of gene expression and the splicing efficiency of introns in maize cells. Mol Gen Genet 225:81–93
Maas C, Laufs J, Grant S, Korfhage C, Werr W (1991) The combination of a novel stimulatory element in the first exon of the maize Shrunken-1 gene with the following intron enhances reporter gene expression up to 1000-fold. Plant Mol Biol 16:199–207
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning: a laboratory manual. Cold Spring Harborg Laboratory, Cold Spring Harbor, NY
Mascarenhas D, Mattler IJ, Pierce DA, Lowe HW (1990) Intron-mediated enhancement of heterologous gene expression in maize. Plant Mol Biol 15:913–920
McElroy D, Rothenberg M, Wu R (1990a) Structural characterization of a rice actin gene. Plant Mol Biol 14:163–171
McElroy D, Rothenberg M, Reece KS, Wu R (1990b) Characterization of the rice (Oryza sativa) actin gene family. Plant Mol Biol 15:257–268
McElroy D, Zhang W, Cao J, Wu R (1990c) Isolation of an efficient actin promoter for use in rice transformation. Plant Cell 2:163–171
Oard JH, Paige D, Dvorak J (1989) Chimeric gene expression using maize intron in cultured cells of breadwheat. Plant Cell Rep 8:156–160
Peterhans A, Datta SK, Datta K, Goodall GJ, Potrykus I, Paszkowski J (1990) Recognition efficiency of Dicotyledoneae-specific promoter and RNA processing signals in rice. Mol Gen Genet 222:361–368
Sleat DA, Gallie DR, Jefferson RA, Bevan MW, Turner PC, Wilson TMA (1987) Characterization of the 5′-leader sequence of tobacco mosaic virus RNA as a general enhancer of translation in vitro. Gene 217:217–225
Spencer TM, Gordon-Kamm WJ, Dames RJ, Start WG, Lemaux PG (1990) Bialaphos selection of stable transformants of maize cell culture. Theor Appl Genet 79:625–631
Tanaka A, Mita S, Ohta S, Kyozuka J, Shimamoto K, Nakamura K (1990) Enhancement of foreign gene expression by a dicot intron in rice but not in tobacco is correlated with an increased level of mRNA and efficient splicing of the intron. Nucleic Acids Res 18:6767–6770
Tereda R, Shimamoto K (1990) Expression of CaMV 35S-GUS gene in transgenic rice plants. Mol Gen Genet 220:389–392
Vasil V, Clancy M, Ferl RJ, Vasil I, Hannah LC (1989) Increased gene expression by the first intron of maize Shrunken-1 locus in grass species. Plant Physiol 91:1575–1579
Zhang W, Wu R (1988) Efficient regeneration of transgenic plants from rice protoplasts and correctly regulated expression of the foreign gene in the plants. Theor Appl Genet 76:835–840
Zhang W, McElroy D, Wu R (1991) Analysis of rice Act1 5′ region activity in transgenic rice plants. Plant Cell 3:1155–1165
Author information
Authors and Affiliations
Additional information
Communicated by E. Meyerowitz
Rights and permissions
About this article
Cite this article
McElroy, D., Blowers, A.D., Jenes, B. et al. Construction of expression vectors based on the rice actin 1 (Act1) 5′ region for use in monocot transformation. Molec. Gen. Genet. 231, 150–160 (1991). https://doi.org/10.1007/BF00293832
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00293832