Summary
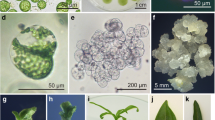
Green mesophyll protoplasts of the dihaploid potato line 198∶2 (Solanum tuberosum L.) were fused with herbicide-bleached mesophyll protoplasts of the dihaploid potato line 67∶9 using a polyethylene glycol protocol. Heterokaryons were identified under a fluorescence microscope using the dual fluorescence of carboxyfluorescein-stained, herbicide-bleached protoplasts and the autofluorescence of green mesophyll protoplasts. About 20% of the protoplasts survived the fusion treatment, and the fusion frequency was 3%–4%. Unfused and fused protoplasts were mass cultured for 6 weeks after which vigorously growing calli were selected and transferred to shoot regeneration medium. Somatic hybrids were identified by a combination of five isozyme markers, and the ploidy level was determined by flow cytometry. Out of 15 calli that regenerated shoots, 6 plants derived from 2 different calli were identified as hexaploid somatic hybrids, while one morphologically deviant plant from a third callus was identified as a mixoploid that had lost some enzyme markers after 4 months of culturing.
Similar content being viewed by others
References
Allicchio R, Antonioli C, Graziani L, Roncavati R, Vannini C (1987) Isozyme variation in leaf-callus regenerated plants of Solanum tuberosum. Plant Sci 53:81–86
Austin S, Baer MA, Helgeson JP (1985a) Transfer of resistance to potato leaf roll virus from Solanum brevidens into Solanum tuberosum by somatic fusion. Plant Sci 39:75–82
Austin S, Baer MA, Ehlenfeldt M, Kazmierczak PJ, Helgeson JP (1985b) Intra-specific fusion in Solanum tuberosum. Theor Appl Genet 71:172–175
Barsby TL, Shepard JF, Kemble RJ, Wong R (1984) Somatic hybridization in the genus Solanum: S. tuberosum and S. brevidens. Plant Cell Rep 3:165–167
Butenko RG, Kuchko AA (1980) Somatic hybridization of Solanum tuberosum L. and Solanum chacoense Bitt. by protoplast fusion. In: Ferenczy L, Farkas GL (eds) Advances in protoplast research. Akad Kiadó, Budapest, pp 293–300
Carlberg I, Glimelius K, Eriksson T (1983) Improved culture ability of potato protoplasts by use of activated charcoal. Plant Cell Rep 2:223–225
Carlberg I, Glimelius K, Eriksson T (1984) Nuclear DNA content during the initiation of callus formation from isolated protoplasts of Solanum tuberosum L. Plant Sci Lett 35:225–230
Carlberg I, Karlsson SB, Eriksson T (1987) Improved culture techniques for potato protoplasts. In: Bajaj YPS (ed) Potato, pp 187–194. Springer, Berlin Heidelberg New York (Biotechnology in Agriculture and Forestry, vol. 3)
Cooper DB, Sears RG, Lookhart GL, Jones BL (1986) Heritable somaclonal variation in gliadin proteins of wheat plants derived from immature embryo callus culture. Theor Appl Genet 71:784–790
Fahleson J, Dixelius J, Sundberg E, Glimelius K (1988) Correlation between flow cytometric determination of nuclear DNA content and chromosome number in somatic hybrids within Brassicaeae. Plant Cell Rep 7:74–77
Fish N, Karp A, Jones MGK (1987) Improved isolation of dihaploid Solanum tuberosum protoplasts and the production of somatic hybrids between dihaploid S. tuberosum and S. brevidens. In Vitro Cell Develop Biol 23:575–580
Foroughi-Wehr B, Wilson HM, Mix G, Gaul H (1977) Monohaploid plants from anthers of dihaploid genotype of Solanum tuberosum L. Euphytica 26:361–367
Glimelius K, Eriksson T, Grafe R, Muller AJ (1978) Somatic hybridization of nitrate reductase deficient mutants of Nicotiana tabacum by protoplast fusion. Physiol Plant 44:273–277
Glimelius K, Djupsjöbacka M, Fellner-Feldegg H (1986) Selection and enrichment of plant protoplast heterokaryons of Brassicaeae by flow sorting. Plant Sci 45:133–141
Hein T, Schieder O (1986) An improved culture method of mechanically isolated heterokaryons of potato. Plant Breed 97:255–260
Hein T, Przewozny T, Schieder O (1983) Culture and selection of somatic hybrids using an auxotrophic cell line. Theor Appl Genet 64:119–122
Helgeson JP, Hunt GJ, Haberlach GT, Austin S (1986) Somatic hybrids between Solanum brevidens and Solanum tuberosum: Expression of a late blight resistance gene and potato leaf roll resistance. Plant Cell Rep 3:212–214
Jacobsen E, Tempelaar MJ, Bijmolt EW (1983) Ploidy levels in leaf callus and regenerated plants of Solanum tuberosum determined by cytophotometric measurements of protoplasts. Theor Appl Genet 65:113–118
Johansson L (1986) Improved methods for induction of embryogenesis in anther cultures of Solanum tuberosum. Potato Res 29:179–191
Menczel L, Nagy F, Kiss ZsR, Maliga P (1981) Streptomycin resistant and sensitive somatic hybrids of Nicotiana tabacum + Nicotiana knightiana: correlation of resistance to N. tabacum plastids. Theor Appl Genet 59:191–195
Oliver JL, Martínez-Zapater JM (1985) A genetic classification of potato cultivars based on allozyme patterns. Theor Appl Genet 69:305–311
Puite KJ, Roest S, Pijnacker LP (1986) Somatic hybrid potato plants after electrofusion od diploid Solanum tuberosum and Solanum phureja. Plant Cell Rep 5:262–265
Shaw CR, Prasad R (1970) Starch gel electrophoresis of enzymes — A compilation of recipes. Biochem Genet 4:297–320
Shepard JF (1980) Abscisic acid-enhanced shoot initiation in protoplast derived calli of potato. Plant Sci Lett 18:327–333
Shields CR, Orton TJ, Stuber CW (1983) An outline of general resource needs and procedures for the electrophoretic separation of active enzymes from plant tissues. In: Tanksley SD, Orton TJ (eds) Isozymes in plant genetics and breeding, part A. Elsevier, Amsterdam, pp 443–468
Soltis DE, Haufler CH, Darrow DC, Gastony GJ (1983) Starch gel electrophoresis of ferns: A complication of grinding buffers, gel and electrode buffers and staining schedules. Am Fern 73:9–27
Sree Ramulu K (1986) Case histories of genetic variability in vitro: potato. In: Vasil IK (ed) Cell culture and somatic cell genetics of plants, vol 3. Academic Press, London New York, pp 449–472
Sundberg E, Glimelius K (1986). A method for the production of interspecific hybrids within Brassiceae via somatic hybridization, using resynthesis of Brassica napus as a model. Plant Sci 43:155–162
Sundberg E, Landgren M, Glimelius K (1987) Fertility and chromosome stability in Brassica napus resynthesised by protoplast fusion. Theor Appl Genet 75:96–104
Uhrig H (1981) Regeneration of protoplasts of dihaploid potato plants bleached by a herbicide (SAN 6706). Mol Gen Genet 181:403–405
Uijtewaal BA, Huigen DJ, Hermsen JGTh (1987a) Production of potato monohaploids (2n=x=12) through prickle pollination. Theor Appl Genet 73:751–758
Uijtewaal BA, Suurs LCJM, Jacobsen E (1987b) Protoplast fusion of monohaploid (2n=x=12) potato clones: identification of somatic hybrids using malate dehydrogenase as a biochemical marker. Plant Sci 51:277–284
Vallejos CE (1983) Enzyme activity staining. In: Tanskley SD, Orton TJ (eds) Isozymes in plant genetics and breeding, part A. Elsevier, Amsterdam, pp 469–516
Wenzel G, Schieder O, Przewozny T, Sopory SK, Melchers G (1979) Comparison of single cell culture derived Solanum tuberosum L. plants and a model for their application in breeding programs. Theor Appl Genet 55:49–55
Wenzel G, Mayer C, Przewozny T, Uhrig H, Schieder O (1982) Incorporation of microspore and protoplast techniques into potato breeding programs. In: Earle ED, Demarly Y (eds) Variability in plants regenerated from tissue culture. Praeger, New York, pp 290–302
Author information
Authors and Affiliations
Additional information
Communicated by J. MacKey
Rights and permissions
About this article
Cite this article
Waara, S., Tegelström, H., Wallin, A. et al. Somatic hybridization between anther-derived dihaploid clones of potato (Solanum tuberosum L.) and the identification of hybrid plants by isozyme analysis. Theoret. Appl. Genetics 77, 49–56 (1989). https://doi.org/10.1007/BF00292315
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00292315