Summary
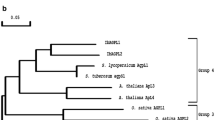
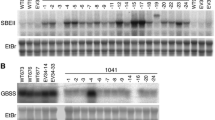
One of the key enzymes involved in the formation of amylopectin, which is the major component of starch, is branching enzyme. A cDNA for potato branching enzyme was cloned by screening a tuber-specific cDNA expression library using an antiserum directed against a denatured preparation of the protein. Complementation of an Escherichia coli strain deficient in branching enzyme was achieved using a construct derived from this clone. Analysis of the expression of the gene in potato revealed a close association with conditions favouring starch biosynthesis. The expression pattern of the gene coding for potato branching enzyme, as analyzed at the mRNA level, closely resembles that of 4GPase S, a gene coding for one of the subunits of ADP-glucose pyrophosphorylase, which is the key regulatory enzyme in the starch biosynthetic pathway. This raises the possibility that enzymes involved in the pathway are coordinately regulated at the transcriptional level.
Similar content being viewed by others
References
Amasino RM (1986) Acceleration of nucleic acid hybridization rate by polyethylene glycol. Anal Biochem 152:304–307
Baecker PA, Greenberg E, Preiss J (1986) Biosynthesis of bacterial glycogen. Primary structure of Escherichia coli 1,4-α-d-glucan: 1,4-α-d-glucan 6-α-d-(1,4-α-d-glucano)-transferase as deduced from the nucleotide sequence of the glgB gene. J Biol Chem 261:8738–8743
Barker SA, Bourne EJ, Wilkinson IA, Peat S (1950) The enzymic synthesis and degradation of starch. Part VII. The mechanism of Q-enzyme action. J Chem Soc 93–99
Bettey M, Smith AM (1990) Nature of the effect of the r locus on the lipid content of embryos of peas (Pisum sativum L.). Planta 180:420–428
Bhattacharyya MK, Smith AM, Ellis THN, Hedley C, Martin C (1990) The wrinkled-seed character of pea described by Mendel is caused by a transposon-like insertion in a gene encoding starch-branching enzyme. Cell 60:115–122
Borovsky D, Smith EE, Whelan WJ (1975) Purification and properties of potato 1,4-α-d-glucan:1,4-α-d-glucan 6-α-(1,4-α-d-glucano)-transferase. Evidence against a dual catalytic function in amylose-branching enzyme. Eur J Biochem 59:615–625
Boyer CD (1985) Soluble starch synthases and branching enzymes from developing seeds of sorghum. Phytochemistry 24:15–18
Boyer CD, Preiss J (1978) Multiple forms of (1,4)-α-d-glucan-6-glucosyl transferase from developing Zea mays L. kernels. Carbohydr Res 61:321–334
Edwards J, Green JH, ap Rees T (1988) Activity of branching enzyme as a cardinal feature of the ra locus in Pisum sativum. Phytochemistry 27:1615–1620
Hahlbrock K, Scheel D (1989) Physiology and molecular biology of phenylpropanoid metabolism. Annu Rev Plant Physiol 40:347–369
Hawker JS, Marschner H, Krauss A (1979) Starch synthesis in developing potato tubers. Plant Physiol 46:25–30
Kauss H (1987) Some aspects of calcium-dependent regulation in plant metabolism. Annu Rev Plant Physiol 38:47–72
Kiel JAKW, Vossen JPMJ, Venema G (1987) A general method for the construction of Escherichia coli mutants by homologous recombination and plasmid segregation. Mol Gen Genet 207:294–301
Kiel JAKW, Elgersma HSA, Beldman G, Vossen JPMJ, Venema G (1989) Cloning and expression of the branching enzyme gene (glgB) from the cyanobacterium Synechococcus sp. PCC7942 in Escherichia coli. Gene 78:9–17
Kiel JAKW, Boels JM, Beldman G, Venema G (1990) Nucleotide sequence of the Synechococcus sp. PCC7942 brancing enzyme gene (glgB): expression in Bacillus subtilis. Gene 89:77–84
Lin TP, Caspar T, Sommerville C, Preiss J (1988) A starch deficient mutant of Arabidopsis thaliana (L.) Heynh lacking ADPglucose pyrophosphorylase activity. Plant Pbysiol 86:1131–1135
Logemann J, Schell J, Willmitzer L (1987) Improved method for the isolation of RNA from plant tissues. Anal Biochem 163:21–26
Matters GL, Boyer CD (1981) Starch synthases and branching enzymes from Pisum sativum. Phytochemistry 20:1805–1809
Mendel G (1865) Versuche über Pflanzen-Hybriden. Verh Naturforsch Ver Brünn 4:3–47
Müller-Rüber BT, Koßmann J, Hannah LC, Willmitzer L, Sonnewald U (1990) One of two different ADP-glucose pyrophosphorylase genes from potato responds strongly to elevated levels of sucrose. Mol Gen Genet 224:136–146
Preiss J (1988) Biosynthesis of starch and its regulation. In: Preiss J (ed) The biochemistry of plants, vol 14. Academic Press, New York, pp 181–254
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: A laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Smith AM (1988) Major differences in isoforms of starch-branching enzyme between developing embryos of round- and wrinkled-seeded peas (Pisum sativum L.). Planta 175:270–279
Smith AM, Neuhaus HE, Stitt M (1990) The impact of decreased activity of starch-branching enzyme on photosynthetic starch synthesis in leaves of wrinkled-seeded peas. Planta 181: 310–315
Svensson B (1988) Regional distant homology between amylases, α-glucosidases and transglucanosylases. FEBS Lett 230:72–76
Turner SR, Barratt DHP, Casey R (1990) The effect of different alleles at the r locus on the synthesis of seed storage proteins in Pisum sativum. Plant Mol Biol 14:793–803
Vos-Scheperkeuter GH, de Wit JG, Ponstein AS, Feenstra WJ, Witholt B (1989) Immunological comparison of the starch branching enzymes from potato tubers and maize kernels. Plant Physiol 90:75–84
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koßmann, J., Visser, R.G.F., Müller-Röber, B. et al. Cloning and expression analysis of a potato cDNA that encodes branching enzyme evidence for co-expression of starch biosynthetic genes. Molec. Gen. Genet. 230, 39–44 (1991). https://doi.org/10.1007/BF00290648
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00290648