Summary
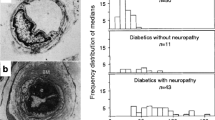
Muscle blood flow and capillary diffusion capacity were determined in 21 Type 1 (insulin-dependent) diabetic patients and in 12 age-matched healthy subjects by measuring the simultaneous clearance of 133xenon and 131iodide from hyperaemic anterior tibial muscle. Blood flow was significantly lower (mean ± SD: 46.7±14.1 versus 59.4±12.9 ml 100 g-1 min-1, p<0.02) and capillary diffusion capacity was significantly greater (mean ± SD: 8.0±2.1 versus 5.9±1.3 mol/min, p<0.005) in the diabetic patients than in the control subjects. Ultrastructural dimensions and density of capillaries in the gastrocnemius muscle of 11 diabetic patients and six control subjects were also studied. Diabetic and control capillaries did not differ in total capillary area. Compared with normal capillaries, the percentage area of basement membrane and the apparent basement membrane thickness were significantly greater (1.21±0.6 versus 0.78±0.2 μ, p<0.05) in diabetic capillaries, while there were no significant differences in luminal, endothelial and pericytial areas. There was no difference in capillary density between the two groups. No correlation was found between basement membrane thickness and capillary diffusion capacity in the diabetic patients. We conclude that the greater capillary diffusion capacity is due to increased permeability of diabetic capillaries, and that the basement membrane is probably not ratelimiting in the transcapillary transport of hydrophilic substances in diabetic subjects.
Article PDF
Similar content being viewed by others

References
Zacks SI, Pegues JJ, Elliott FA (1962) Interstitial muscle capillaries in patients with diabetes mellitus: a light and electron microscope study. Metabolism 11:381–393
Siperstein MD, Unger RH, Madison LL (1968) Studies of capillary basement membranes in normal subjects, diabetic, and prediabetic patients. J Clin Invest 47:1973–1999
Vracko R (1970) Skeletal muscle capillaries in diabetics: a quantitative analysis. Circulation 41:271–284
Kilo C, Vogler N, Williamson JR (1972) Muscle capillary basement membrane changes related to aging and to diabetes mellitus. Diabetes 21:881–905
Danowski TS, Fisher ER, Khurana RC, Nolan S, Stephan T (1972) Muscle capillary basement membrane in juvenile diabetes mellitus. Metabolism 21:1125–1132
Williamson JR, Kilo C (1977) Current status of capillary basement-membrane disease in diabetes mellitus. Diabetes 26:65–73
Gundersen HJG, Østerby R, Lundbaek K (1978) The basement membrane controversy. Diabetologia 15:361–363
Siperstein MD, Feingold KR, Bennett PH (1978) Hyperglycaemia and diabetic microangiopathy. Diabetologia 15:365–367
Jerusalem F, Rakusa M, Engel AG, MacDonald RD (1974) Morphometric analysis of skeletal muscle capillary ultrastructure in inflammatory myopathies. J Neurol Sci 23:391–402
Longhurst J, Capone RJ, Zelis R (1975) Evaluation of skeletal muscle capillary basement membrane thickness in congestive heart failure. Chest 67:195–198
Mäkitie J (1977) Skeletal muscle capillaries in intermittent claudication. Arch Pathol Lab Med 101:500–503
McMillan DE (1978) Diabetic angiopathy — its lessons in vascular pathology. Am Heart J 96:401–406
Parving HH, Rossing N (1973) Simultaneous determination of the transcapillary escape rate of albumin and IgG in normal and long-term juvenile diabetic subjects. Scand J Clin Lab Invest 32:239–244
Trap-Jensen J, Alpert JS, Del Rio G, Lassen NA (1967) Capillary diffusion capacity for sodium in skeletal muscle in longterm juvenile diabetes mellitus. Acta Med Scand Suppl 476:135–146
Trap-Jensen J, Lassen NA (1968) Increased capillary diffusion capacity for small ions in skeletal muscle in long-term diabetics. Scand J Clin Lab Invest 21:116–122
Trap-Jensen J (1970) Increased capillary permeability to 131Iodide and 51CrEDTA in exercising forearm of long-term diabetics. Clin Sci 39:39–49
Alpert JS, Coffman JD, Balodimos MC, Koncz L, Soeldner JS (1972) Capillary permeability and blood flow in skeletal muscle of patients with diabetes mellitus and genetic prediabetes. N Engl J Med 286:454–460
Munck O, Lindbjerg IF, Binder C, Lassen NA, Trap-Jensen J (1966) Skeletal muscle blood flow in diabetic patients determined by intramuscular injection of Xenon-133. Diabetes 15:323–326
Christensen JN (1968) Muscle blood flow, measured by Xenon-133 and vascular calcifications in diabetics. Acta Med Scand 183:449–154
Leinonen H, Salminen S, Peltokallio P (1978) Capillary permeability and maximal blood flow in skeletal muscle in athletes and non-athletes measured by local clearances of 133Xe and 131I-. Scand J Clin Lab Invest 38:223–227
Lassen NA, Trap-Jensen J (1968) Theoretical considerations on measurement of capillary diffusion capacity in skeletal muscle by the local clearance method. Scand J Clin Lab Invest 21:108–115
Vracko R (1970) Skeletal muscle capillaries in nondiabetics: A quantitative analysis. Circulation 41:285–297
Brodal P, Ingjer F, Hermansen L (1977) Capillary supply of skeletal muscle fibers in untrained and endurance-trained men. Am J Physiol 232:H705–712
Poulsen HL, Nielsen SL (1976) Water filtration of the forearm in short- and long-term diabetes mellitus. Diabetologia 12:437–440
Katz MA, Janjan N (1978) Forearm hemodynamics and responses to exercise in middle-aged adult-onset diabetic patients. Diabetes 27:726–731
Norton WL (1970) Comparison of the microangiopathy of systemic lupus erythematosus, dermatomyositis, scleroderma, and diabetes mellitus. Lab Invest 22:301–308
Renkin EM (1977) Multiple pathways of capillary permeability. Circ Res 41:735–743
Leinonen H, Juntunen J, Somer H, Rapola J (1979) Capillary circulation and morphology in Duchenne muscular dystrophy. Eur Neurol 18:249–254
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leinonen, H., Matikainen, E. & Juntunen, J. Permeability and morphology of skeletal muscle capillaries in type 1 (insulin-dependent) diabetes mellitus. Diabetologia 22, 158–162 (1982). https://doi.org/10.1007/BF00283744
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00283744