Summary
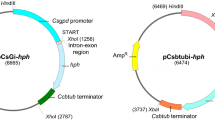
A transformation system for Ophiostoma ulmi (Buism.) Nannf. was developed and analyzed. Protoplasts were generated from actively budding yeastlike cells by digestion with NovoZym 234 in MgSO4 after pretreatment with 2-mercaptoethanol. Protoplast regeneration was most efficient when 0.6 M sucrose was used as the osmoticum. Several plasmids containing fusions between fungal promoters and a bacterial gene for hygromycin phosphotransferase successfully transformed O. ulmi to hygromycin resistance. One of these vectors, pPS57, which contains a promoter for isopenicillin N synthetase from Penicillium chrysogenum, consistently conferred the greatest resistance to hygromycin. Linearization of the vector and inclusion of 2-mercaptoethanol in the transformation reaction resulted in enhanced transformation efficiency. Approximately 4 × 103 transformants/μg DNA per 107 protoplasts were obtained using the optimized procedure. Southern hybridization after alternating field and standard electrophoresis suggested random insertion of tandem repeats (some greater than 250 kb) into the fungal chromosomes. Antibiotic resistance was stable through mitosis. However, expression of the transforming DNA after meiosis was highly variable.
Similar content being viewed by others
References
Anonymous (1989) New varieties of elm raise hope of rebirth for devastated tree. New York Times, Dec 5, p 21
Asina S, Hubbes M (1980) Compatibility in Ceratocystis ulmi. Eur J For Pathol 10:201–208
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1989) Current protocols in molecular biology unit 2.5B, pulsed-field gel electrophoresis. Wiley, Toronto
Bernier L (1988) Induction, characterization, and mapping of mutations in Ophiostoma ulmi, the causal agent of Dutch elm disease. PhD thesis, University of Toronto
Bernier L, Hubbes M (1990a) Mutations in Ophiostoma ulmi induced by N-methyl-N′-nitro-N-nitrosoguanidine. Can J Bot 68:225–231
Bernier L, Hubbes M (1990b) Meiotic analysis of induced mutations in Ophiostoma ulmi. Can J Bot 68:232–235
Binninger DM, Skrzynia C, Pukilla PJ, Casselton LA (1987) DNA-mediated transformation of the basidiomycete fungus Coprinus cinereus. EMBO J 6:835–840
Brasier CM (1981) Laboratory investigation of Ceratocystis ulmi. In: Stipes RJ, Campana RJ (eds) Compendium of elm diseases. American Phytopathological Society, St. Paul, Minn., pp 76–79
Brasier CM (1990) China and the origins of Dutch elm disease: an appraisal. Plant Pathol 39:5–16
Brasier CM, Gibbs JN (1976) Inheritance of pathogenicity and cultural characters in Ceratocystis ulmi: hybridisation of aggressive and non-aggressive strains. Ann Appl Biol 83:31–37
Cambareri EB, Jensen BC, Schabtach E, Selker ELF (1989) Repeated-induced G-C to A-T mutations in Neurospora. Science 244:1571–1575
Churchill ACL, Ciuffetti LM, Hansen DR, Van Etten HD, Van Alfen NK (1990) Transformation of the fungal pathogen Cryphonectria parasitica with a variety of heterologous plasmids. Curr Genet 17:25–31
Cooley RN, Shaw RK, Franklin FCH, Caten CE (1988) Transformation of the phytopathogenic fungus Septoria nodorum to hygromycin resistance. Curr Genet 13:383–389
de Bruijn FG, Lupski JR (1984) The use of transposon Tn5 mutagenesis in the rapid generation of correlated physical and genetic maps of DNA segments cloned into multicopy plasmids — A review. Gene 27:131–149
DeHoog GS, Scheffer RJ (1984) Ceratocystis versus Ophiostoma: a reappraisal. Mycologia 76:292–299
Fincham JRS (1989) Transformation in Fungi. Microbiol Rev 53:148–170
Fincham JRS, Day PR, Radford A (1979) Fungal genetics. University of California Press, Los Angeles
Harris JL, Taber WA (1970) Influence of certain nutrients and light on growth and morphogenesis of the synnema of Ceratocystis ulmi. Mycologia 62:152–170
Holmes FW (1977) Distinction between sex and compatibility in Ceratocystis ulmi, as shown by unisexual male cultures. Mycologia 64:1149–1161
Hubbes M (1988) Pathogenic virulence and host resistance in Dutch elm disease. Naturalist Can 115:157–161
Huntley GD (1981) The elm — A resurgent resource or a persistent problem? In: Kondo ES, Hiratsuka Y, Denyer WBG (eds) Dutch elm disease symposium and workshop. Manitoba Department of Natural Resources, Forest Production and Dutch Elm Disease Branch, Winnipeg, Manitoba, Canada
Kistler HC, Benny UK (1988) Genetic transformation of the fungal plant wilt pathogen, Fusarium oxysporum. Curr Genet 13:145–149
Kulkarni RK, Nickerson KW (1981) Nutritional control of dimorphism in Ceratocystis ulmi. Exp Mycol 5:148–154
Lanier GN (1989) Trap trees for control of Dutch elm disease. J Arboriculture 15:105–111
Lester DT (1978) Exploiting host resistance. In: Sinclair WA, Campana RJ (eds) Dutch elm disease: Perspective after 60 years. Northeast Regional Research Publication, Search Agriculture Vol. 8, pp 39–42
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning; a laboratory manual. Cold Spring Harbor Laboratory Press, New York
Marshal MR, Hindal DF, MacDonald WL (1982) Production of perithecia in culture of Ceratocystis ulmi. Mycologia 74:376–381
Mellon FM, Casselton LA (1988) Transformation as a method of increasing gene copy number and gene expression in the basidiomycete fungus Coprinus cinereus. Curr Genet 14:451–456
Orbach MJ, Vollrath D, Davis RW, Yanofsky C (1988) An electrophoretic karyotype of Neurospora crassa. Mol Cell Biol 8:1469–1473
Parsons KA, Chumley FG, Valent B (1987) Genetic transformation of the fungal pathogen responsible for rice blast disease. Proc Natl Acad Sci USA 84:4161–4165
Picard M, Debuchy R, Julien J, Brygoo Y (1987) Transformation by integration in Podospora anserina. II. Targeting to the resident locus with cosmids and instability of the transformants. Mol Gen Genet 210:129–134
Queener SW, Ingolia TD, Skatrud PL, Chapman JL, Kaster KR (1985) A system for genetic transformation of Cephalosporium acremonium. Microbiology 1985. American Society for Microbiology, pp 468–472
Reed KC, Mann DA (1985) Rapid transfer of DNA from agarose gels to nylon membranes. Nucleic Acids Res 13:7207–7222
Schafer W, Straney D, Ciuffetti L, Van Etten HD, Yoder OC (1989) One enzyme makes a fungal pathogen, but not a saprophyte, virulent on a new host pathogen. Science 246:247–249
Selker EU, Garret PW (1988) DNA sequence duplications trigger gene inactivation in Neurospora crassa. Proc Natl Acad Sci USA 85:6870–6874
Selker EU, Cambareri EB, Jensen BC, Haack KR (1987) Rearrangement of duplicated DNA in the specialized cells of Neurospora. Cell 51:741–752
Skatrud PL, Queener SW, Carr LG, Fisher DL (1987) Efficient integrative transformation of Cephalosporium acremonium. Curr Genet 12:337–348
Smalley EB (1978) Systemic chemical treatment of trees for protection and therapy. In: Sinclair WA, Campana RJ (eds) Dutch elm disease: Perspective after 60 years. Northeast Regional Research Publication, Search Agriculture Vol. 8, pp. 34–39
Stipes RJ, Campana RJ (1981) Compendium of elm diseases. American Phytopathology Society, St. Paul, Minn
Svircev AM, Jeng RS, Hubbes M (1988) Detection of cerato-ulmin on aggressive isolates of Ophiostoma ulmi by immunocytochemistry and scanning electron microscopy. Phytopathology 78:322–327
Szabo G, Schablik M (1982) Behaviour of DNA-induced inositol independent transformants of Neurospora crassa in sexual crosses. Theor Appl Genet 61:171–175
Takai S (1974) Pathogenicity and cerato-ulmin production in Ceratocystis ulmi. Nature 252:124–126
Tilburn J, Scazzochio C, Taylor GG, Zabicky-Zissman JH, Lockington RA, Davies RW (1983) Transformation by integration in Aspergillus nidulans. Gene 26:205–221
Turgeon BG, Garber RC, Yoder OC (1985) Transformation of the fungal maize pathogen Cochhobolus heterostrophus using the Aspergillus nidulans amdS gene. Mol Gen Genet 201:450–453
Turgeon BG, Garber RC, Yoder OC (1987) Development of a fungal transformation system based on selection of sequences with promoter activity. Mol Cell Biol 7:3297–3305
Vollmer SJ, Yanofsky C (1986) Efficient cloning of genes of Neurospora crassa. Proc Natl Acad Sci USA 83:4869–4873
Wang J, Holden DW, Leong SA (1988) Gene transfer system for the phytopathogenic fungus Ustilago maydis. Proc Natl Acad Sci USA 85:865–869
Webber JF, Mitchell AG, Smith F (1986) Linkage of the genes determining mating type and fungicide tolerance in Ophiostoma ulmi. Plant Pathol 35:512–516
Yang D, Jeng RS, Hubbes M (1989) Mansonone accumulation in elm callus induced by elicitors of Ophiostoma ulmi; and general properties of elicitors. Can J Bot 67:3490–3497
Zolan M, Pukkila P (1986) Inheritance of DNA methylation in Coprinus cinereus. Mol Cell Biol 6:195–200
Author information
Authors and Affiliations
Additional information
Communicated by C.A. van den Hondel
Rights and permissions
About this article
Cite this article
Royer, J.C., Dewar, K., Hubbes, M. et al. Analysis of a high frequency transformation system for Ophiostoma ulmi, the causal agent of Dutch elm disease. Molec. Gen. Genet. 225, 168–176 (1991). https://doi.org/10.1007/BF00282655
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00282655