Abstract
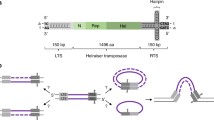
Plasmids carrying two IS30 elements in the same orientation, as in the composite transposon Tn2706, are structurally unstable in Escherichia coli. A primary segregation product is formed by site-specific deletion of the sequences carried between the two IS30 elements. The resulting covalently closed replicon carries the two IS30 elements as tandem repeats separated by only 2 bp. This (IS30)2 structure is extremely unstable, but it can nevertheless be isolated on its vector plasmid and, after purification, can be reintroduced into host cells by transformation. Among the descendants of transformants of recA − bacteria, replicated copies of the introduced (IS30)2 structure are still present, together with various kinds of segregation products which provide evidence for the efficient generation of DNA rearrangements. Most abundant is the product of another site-specific recombination between two identical ends of the IS30 elements involved, which results in the presence of just one intact IS30 on the plasmid. Apart from this, and depending on the presence of appropriate targets for IS30 transposition, various transposition products of (IS30)2 are also seen. Intramolecular reactions lead to DNA inversions and deletions with breakpoints other than IS30 ends. In intermolecular reactions inverse transposition occurs at high frequency and one also obtains simple transposition and cointegration. A mutational study revealed the requirement in cis of one intact IS30 transposase gene and of both proximal ends of the two IS30 elements concerned not only for the formation of (IS30)2, but also for its further rearrangement reactions, including the efficient formation of site-specific deletions. A model is proposed, which postulates that (IS30)2 intermediates play a key role in IS30 transposition pathways in which the formation of (IS30)2 may be rate-limiting. Once this structure is formed, it gives rise to a burst of transpositional rearrangements in the subclone carrying (IS30)2. Evolutionary implications of these findings are discussed.
Similar content being viewed by others
References
Ahmed A (1986) Evidence for replicative transposition of Tn5 and Tn9. J Mol Biol 191:75–84
Benjamin HW, Kleckner N (1989) Intramolecular transposition by Tn10. Cell 59:373–383
Berg DE (1983) Structural requirement for IS50-mediated gene transposition. Proc Natl Acad Sci USA 80:792–796
Berg DE (1989) Transposon Tn5. In: Berg DE, Howe MM (eds) Mobile DNA. American Society for Microbiology, Washington DC, pp 185–210
Birkenbihl RP, Vielmetter W (1989) Complete maps of IS1, IS2, IS3, IS4, IS5, IS30 and IS150 locations in Escherichia coli K12. Mol Gen Genet 220:147–153
Caparon MG, Scott JR (1989) Excision and insertion of the conjugative transposon Tn916 involves a novel recombination mechanism. Cell 59:1027–1034
Caspers P, Dalrymple B, Iida S, Arber W (1984) IS30, a new insertion sequence of Escherichia coli K12. Mol Gen Genet 196:68–73
Chang ACJ, Cohen SN (1978) Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J Bacteriol 134:1141–1156
Dalrymple B (1987) Novel rearrangements of IS30-carrying plasmids leading to the reactivation of gene expression. Mol Gen Genet 207:413–420
Dalrymple B, Arber W (1985) Promotion of RNA transcription on the insertion element IS30 of E. coli K12. EMBO J 4:2687–2693
Dalrymple B, Arber W (1986) The characterization of terminators of RNA transcription of IS30 and an analysis of their role in IS element-mediated polarity. Gene 44:1–10
Dalrymple B, Caspers P, Arber W (1984) Nucleotide sequence of the procaryotic mobile genetic element IS30. EMBO J 3:2145–2149
Dente L, Cesareni G, Cortese R (1983) pEMBL: a new family of single stranded plasmids. Nucleic Acids Res 11:1645–1655
Galas DJ, Chandler M (1989) Bacterial insertion sequences. In: Berg DE, Howe MM (eds) Mobile DNA. American Society for Microbiology, Washington DC, pp 109–162
Haniford DB, Benjamin HW, Kleckner N (1991) Kinetic and structural analysis of a cleaved donor intermediate and a strand transfer intermediate in Tn10 transposition. Cell 64:171–179
Hirschel BJ, Galas DJ, Chandler M (1982) Cointegrate formation by Tn5, but not transposition, is dependent on recA. Proc Natl Acad Sci USA 79:4530–4534
Lichens-Park A, Syvanen M (1988) Cointegrate formation by IS50 requires multiple donor molecules. Mol Gen Genet 211:244–251
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning, a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Messing J (1979) A multipurpose cloning system based on a single-stranded DNA bacteriophage M13. Recombinant Tech Bull 2:43–48
Miller J (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Morisato D, Kleckner N (1984) Transposase promotes double strand breaks and single strand joints at Tn10 termini. Cell 39:181–190
Morisato D, Kleckner N (1987) Tn10 transposition and circle formation in vitro. Cell 51:101–111
Muster CJ, Shapiro JA (1980) Recombination involving transposable elements: On replicon fusion. Cold Spring Harbor Symp Quant Biol 45:239–242
Reimmann C, Haas D (1987) Mode of replicon fusion mediated by the duplicated insertion sequence IS21 in Escherichia coli. Genetics 115:619–625
Reimmann C, Hass D (1990) The intA gene of insertion sequence IS21 is essential for cleavage at the inner 3′ ends of randomly repeated IS21 elements in vitro. EMBO J 9:4055–4063
Reimmann C, Moore R, Little S, Savioz A, Willetts NS, Haas D (1989) Genetic structure, function and regulation of the transposable element IS21. Mol Gen Genet 215:416–424
Rose AM, Snutch TP (1984) Isolation of the closed circular form of the transposable element Tc1 in Caenorhabditis elegans. Nature 311:485–486
Ruan K, Emmons W (1984) Extrachromosomal copies of transposon Tc1 in the nematode Caenorhabditis elegans. Proc Natl Acad Sci USA 81:4018–4022
Russel M, Kidd S, Kelley MR (1986) An improved filamentous helper phage for generating single-stranded plasmid DNA. Gene 45:333–338
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Scott JN, Kirchman PA, Caparon MG (1988) An intermediate in transposition of the conjugative transposon Tn916. Proc Natl Acad Sci USA 85:4809–4813
Spielmann-Ryser J, Moser M, Kast P, Weber H (1991) Factors determining the frequency of plasmid cointegrate formation mediated by insertion sequence IS3 from Escherichia coli. Mol Gen Genet 226:441–448
Stalder R, Arber W (1989) Characterization of in vitro constructed IS30-flanked transposons. Gene 76:187–193
Stalder R, Caspers P, Olasz F, Arber W (1990) The N-terminal domain of the IS30 transposase is able to interact specifically with the terminal inverted repeats of the element. J Biol Chem 265:3757–3762
Sundaresan V, Freeling M (1987) An extrachromosomal form of the Mu transposons of maize. Proc Natl Acad Sci USA 84:4924–4928
Umeda M, Ohtsubo E (1990) Mapping of insertion element IS30 in the Escherichia coli K12 chromosome. Mol Gen Genet 222:317–322
Vieira J, Messing J (1982) The pUC plasmids, and M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene 19:259–268
Weinert TA, Derbyshire KM, Hughson FM, Grindley ND (1984) Replicative and conservative transpositional recombination of insertion sequences. Cold Spring Harbor Symp Quant Biol 49:251–260
Weinert TA, Schaus NA, Grindley ND (1983) Insertion sequence duplication in transpositional recombination. Science 222:755–765
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Author information
Authors and Affiliations
Additional information
Communicated by H. Hennecke
Rights and permissions
About this article
Cite this article
Olasz, F., Stalder, R. & Arber, W. Formation of the tandem repeat (IS30)2 and its role in IS30-mediated transpositional DNA rearrangements. Molec. Gen. Genet. 239, 177–187 (1993). https://doi.org/10.1007/BF00281616
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00281616