Abstract
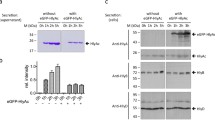
Escherichia coli hemolysin (HlyA) is secreted by a specific export machinery which recognizes a topogenic secretion signal located at the C-terminal end of HlyA. This signal sequence has been variously defined as comprising from 27 to about 300 amino acids at the C-terminus of HlyA. We have used here a combined genetic and immunological approach to select for C-terminal HlyA peptides that are still secretion-component. A deletion library of HlyA mutant proteins was generated in vitro by successive degradation of hy1A from the 5′ end with exonuclease III. Secretion competence was tested by immunoblotting of the supernatant of each clone with an antiserum raised against a C-terminal portion of hemolysin. It was found that the hemolysin secretion system has no apparent size limitation for HlyA proteins over a range from 1024 to 62 amino acids. The smallest autonomously secretable peptide isolated in this selection procedure consists of the C-terminal 62 amino acids of HlyA. This sequence is shared by all secretion-competent, truncated HlyA proteins, which suggests that secretion of the E.coli hemolysin is strictly post-translational. The capacity of the hemolysin secretion machinery was found to be unsaturated by the steady-state level of its natural HlyA substrate and large amounts of truncated HlyA derivatives could still be secreted in addition to full-length HlyA.
Similar content being viewed by others
References
Cleveland DW (1983) Peptide mapping in one dimension by limited proteolysis of sodium dodecyl sulfate-solubilized proteins. Methods Enzymol 96:222–229
Coote JG (1992) Structural and functional relationships among the RTX toxin determinants of gram-negative bacteria. FEMS Microbiol Rev 88:137–162
Delepelaire P, Wandersman C (1990) Protein secretion in gram-negative bacteria: the extracellular metalloprotease B from Erwinia chrysanthemi contains a C-terminal secretion signal analogous to that of Escherichia coli α-hemolysin. J Biol Chem 265:17:118–125
Economou AE, Hamilton WDO, Johnston AWB, Downie JA (1990) The rhizobium modulation gene nodO encodes a Ca2+binding protein that is exported without an N-terminal cleavage and is homologous to hemolysin and related proteins. EMBO J 9:349–354
Fath MJ, Skvirsky RC, Kolter R (1991) Functional complementation between bacterial MDR-like export systems: colicin V, α-hemolysin, and Erwinia protease. J Bacteriol 173:7549–7556
Felmlee T, Welch RA (1988) Alterations of amino acid repeats in the Escherichia coli hemolysin affect cytolytic activity and secretion. Proc Natl Acad Sci USA 85:5269–5273
Felmlee T, Pellett S, Lee E-Y, Welch RA (1985a) Escherichia coli hemolysin is released extracellularly without cleavage of a signal peptide. J Bacteriol 163:88–93
Felmlee T, Pellett S, Welch RA (1985b) Nucleotide sequence of an Escherichia coli chromosomal hemolysin. J Bacteriol 163:94–105
Ferenci T, Silhavy TJ (1987) Sequence information required for protein translocation from the cytoplasm. J Bacteriol 169:5339–5342
Gentschev I, Hess J, Goebel W (1990) Change in the cellular localization of alkaline phosphatase by alteration of its carboxyterminal sequence. Mol Gen Genet 222:211–216
Guzzo J, Duong F, Wandersman C, Murgier M, Lazdunski A (1991) The secretion genes of Pseudomonas aeruginosa alkaline protease are functionally related to those of Erwinia chrysanthemi proteases and Escherichia coli α-hemolysin. Mol Microbiol 5:447–453
Hanke C, Hess J, Schumacher G, Goebel W (1992) Processing by OmpT of fusion proteins carrying the HlyA transport signal during secretion by the Escherichia coli hemolysin transport system. Mol Gen Genet 233:42–48
Härtlein M, Schiessl S, Wagner W, Rdest U, Kreft J, Goebel W (1983) Transport of hemolysin by Escherichia coli. J Cell Biochem 22:87–97
Hawkes R, Niday E, Gordon J (1982) A dot-immunobinding assay for monoclonal and other antibodies. Anal Biochem 119:142–147
Henikoff S (1987) Unidirectional digestion with exonuclease III in DNA sequence analysis. Methods Enzymol 155:156–165
Hess J, Wels W, Vogel M, Goebel W (1986) Nucleotide sequence of a plasmid-encoded hemolysin determinant and its comparison with a corresponding chromosonal hemolysin sequence. FEMS Microbiol Lett 34:1–11
Hess J, Gentschev I, Goebel W, Jarchau T (1990) Analysis of the hemolysin secretion system by PhoA-HlyA fusion proteins. Mol Gen Genet 244:201–208
Kenny B, Haig R, Holland IB (1991) Analysis of the hemolysin transport process through the secretion from Escherichia coli of PCM, CAT or β-galactosidase fused to the HlyA C-terminal domain. Mol Microbiol 510:2557–2568
Koronakis V, Koronakis E, Hughes C (1989) Isolation and analysis of the C-terminal signal directing export of Escherichia coli hemolysin protein across both bacterial membranes. EMBO J 8:595–605
Kyhse-Anderson J (1984) Electroblotting of multiple gels: a simple apparatus without buffer tank for rapid transfer of proteins from polyacrylamide to nitrocellulose. J Biochem Biophys Methods 10:203–209
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Letoffe S, Wandersman C (1992) Secretion of CyaA-PrtB and HlyA-PrtB fusion proteins in Escherichia coli: involvement of the glycine-rich repeat domain of Erwinia chrysanthemi protease B. J Bacteriol 174:4920–4927
Letoffe S, Delepelaire P, Wandersman C (1991) Cloning and expression in Escherichia coli of the Serratia marcescens metalloprotease gene: secretion of the protease from E. coli in the presence of the Erwinia chrysanthemi protease secretion functions. J Bacteriol 173:2160–2166
Ludwig A, Goebel W (1991) Genetic determinants of cytolytic toxins from gram-negative bacteria. In: Alouf JE, Freer JH (eds) Sourcebook of bacterial protein toxins. Academic Press, London, pp 117–146
Mackman N, Nicaud J-M, Gray L, Holland IB (1985) Genetical and functional organisation of the Escherichia coli haemolysin determinant 2001. Mol Gen Genet 201:282–288
Mackman N, Baker K, Gray L, Haigh R, Nicaud J-M, Holland IB (1987) Release of a chimeric protein into the medium from Escherichia coli using the C-terminal secretion signal of haemolysin. EMBO J 6:2835–2841
Nicaud J-M, Mackman N, Gray L, Holland IB (1986) The C-terminal 23 kDa peptide of E. coli haemolysin 2001 contains all the information necessary for its secretion by the haemolysin (Hly) export machinery. FEBS Lett 204:331–335
Olmsted JB (1986) Analysis of cytoskeletal structures using blotpurified monospecific antibodies. Methods Enzymol 134:467–472
Oliver D-B (1987) Periplasm and protein secretion. In: Neidhardt, FC (ed) Escherichia coli and Salmonella typhimurium: cellular and molecular biology. American Society for Microbiology, Washington, DC, pp 56–69
Pellett S, Boehm DF, Synder IS, Rowe G, Welch RA (1990) Characterization of monoclonal antibodies against the Escherichia coli hemolysin. Infect Immun 58:822–827
Pugsley AP (1988) Protein secretion across the outer membrane of gram-negative bacteria. In: Das RC, Robbins PW (eds) Protein transfer and organelle biogenesis. Academic Press, San Diego, pp 607–652
Wagner W, Vogel M, Goebel W (1983) Transport of hemolysin across the outer membrane of Escherichia coli requires two functions. J Bacteriol 154:200–210
Wandersman C, Delepelaire P (1990) Tol C, an Escherichia coli outer membrane protein required for hemolysin secretion. Proc Natl Acad Sci USA 87:4776–4780
Zagursky RJ, Baumeister K, Lomax N, Berman ML (1985) Rapid and easy sequencing of large linear double-stranded DNA and supercoiled plasmid DNA. Gene Anal Technol 2:89–94
Author information
Authors and Affiliations
Additional information
Communicated by E. Bautz
Rights and permissions
About this article
Cite this article
Jarchau, T., Chakraborty, T., Garcia, F. et al. Selection for transport competence of C-terminal polypeptides derived from Escherichia coli hemolysin: the shortest peptide capable of autonomous HIyB/HIyD-dependent secretion comprises the C-terminal 62 amino acids of HlyA. Molec. Gen. Genet. 245, 53–60 (1994). https://doi.org/10.1007/BF00279750
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00279750