Summary
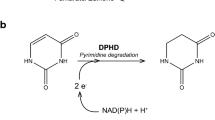
A 3.7 kb DNA fragment of yeast chromosome IV has been sequenced that contains the SFA gene which, when present on a multi-copy plasmid in Saccharomyces cerevisiae, confers hyper-resistance to formaldehyde. The open reading frame of SFA is 1158 by in size and encodes a polypeptide of 386 amino acids. The predicted protein shows strong homologies to several mammalian alcohol dehydrogenases and contains a sequence characteristic of binding sites for NAD. Overexpression of the SFA gene leads to enhanced consumption of formaldehyde, which is most probably the reason for the observed hyper-resistance phenotype. In sfa:LEU2 disruption mutants, sensitivity to formaldehyde is correlated with reduced degradation of the chemical. The SFA gene shares an 868 by divergent promoter with UGX2 a gene of yet unknown function. Promoter deletion studies with a SFA promoter-lacZ gene fusion construct revealed negative interference on expression of SFA by upstream sequences. The upstream region between positons − 145 and − 172 is totally or partially responsible for control of inducibility of SFA by chemicals such as formaldehyde (FA), ethanol and methyl methanesulphonate. The 41 kDa SFA-encoded protein was purified from a hyper-resistant transformant; it oxidizes long-chain alcohols and, in the presence of glutathione, is able to oxidize FA. SFA is predicted to code for a long-chain alcohol dehydrogenase (glutathione-dependent formaldehyde dehydrogenase) of the yeast S. cerevisiae.
Similar content being viewed by others
References
Ausubel FM, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1988) Current protocols in molecular biology. Wiley, New York
Beck CF, Warren AJ (1988) Divergent promoters, a common form of gene organization. Microbiol Rev 52:318–326
Brandford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of protein-dye binding. Anal Biochem 72:248–254
Crabb DW, Edenberg HJ (1986) Complete amino acid sequence of rat liver alcohol dehydrogenase deduced from the cDNA sequence. Gene 48:287–291
Dagert M, Ehrlich SD (1979) Prolonged incubation in calcium chloride improves the competence of E. coli cells. Gene 6:23–28
Del Sal G, Manfioletti G, Schneider C (1988) A one tube plasmid DNA mini-preparation suitable for sequencing. Nucleic Acids Res 16:9878
Dobson MJ, Tuite MF, Roberts NA, Kingsman AJ, Kingsman SM (1982) Conservation of high efficiency promotor sequences in Saccharomyces cerevisiae. Nucleic Acids Res 10:2625–2637
Edenberg HJ, Zhang K, Fong K, Bosron WF, Li T-K (1985) Cloning and sequencing of cDNA encoding the complete mouse liver alcohol dehydrogenase. Proc Natl Acad Sci USA 82:2262–2266
Fleer R, Brendel M (1979) Formation and fate of cross-links induced by polyfunctional anticancer drugs in yeast. Mol Gen Genet 176:41–52
Gömpel-Klein P, Mack M, Brendel M (1989) Molecular characterization of the two genes SNQ and SFA that confer hyperresistance to 4-nitroquinoline-N-oxide and formaldehyde in Saccharomyces cerevisiae. Curr Genet 16:65–74
Gutheil WG, Holmquist B, Vallee BL (1992) Purification, characterization, and partial sequence of the glutathione-dependent formaldehyde dehydrogenase from Escherichia coli: a class III alcohol dehydrogenase. Biochemistry 31:475–481
Hames BD, Rickwood D (1981) Gel electrophoresis of protein: a practical approach. IRL Press, Oxford and Washington, DC, pp 1–91
Hill JE, Myers AM, Körner TJ, Tzagoloff A (1986) Yeast/E. coli shuttle vectors with unique restriction sites. Yeast 2:163–167
Holmquist B, Vallee BL (1991) Human liver class III alcohol and glutathione-dependent formaldehyde dehydrogenase are the same enzyme. Biochem Biophys Res Commun 178:1371–1377
Hoog JO, von Bahr-Lindstrom H, Heden LO, Holmquist B, Larsson K, Hempel J, Vallee BL, Jornvall H (1987) Structure of the class II enzyme of human liver alcohol dehydrogenase: combined cDNA and protein sequence determination of the pi subunit. Biochemistry 26:1926–2932
Ito H, Fukuda J, Murato K, Kimura A (1983) Transformation of intact yeast cells treated with alkali cations. J Bacteriol 153:163–168
Jornvall H (1977) The primary structure of yeast alcohol dehydrogenase. Eur J Biochem 72:425–442
Julia P, Pares X, Jornvall H (1988) Rat liver alcohol dehydrogenase of class III; primary structure, functional consequences and relationships to other alcohol dehydrogenases. Eur J Biochem 172:73–83
Kaiser R, Holmquist B, Hempel J, Vallee BL, Joernvall H (1988) Class III human liver alcohol dehydrogenase: a novel structural type equidistantly related to the class I and class II enzymes. Biochemistry 27:1132–1140
Koivusalo M, Baumann M, Uotila L (1989) Evidence for identity of glutathione-dependent formaldehyde dehydrogenase and class III alcohol dehydrogenase. FEBS Lett 257:105–109
Kyte J, Doolittle RF (1982) A simple method for displaying the hydropathic character of a protein. J Mol Biol 157:105–132
Lipman DJ, Pearson WR (1985) Rapid protein sequence similarity searches. Science 227:1435–1441
Mack M, Gömpel-Klein P, Haase E, Hietkamp J, Ruhland A, Brendel M (1988) Genetic characterization of hyperresistance to formaldehyde and 4-nitroquinoline-N-oxide in the yeast Saccharomyces cerevisiae. Mol Gen Genet 211:260–265
Mason RP, Sanders KM (1986) Formaldehyde metabolism by Escherichia coli. Detection by in vivo 13C NMR spectroscopy of S-(hydroxymethyl)glutathione as a transient intracellular intermediate. Biochemistry 25:4504–4507
Miller JM (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory, Cold Spring Harbor, New York
Rodriguez RL, Tait RC (1983) Recombinant DNA techniques: An introduction. Addison-Wesley, London
Rose ZB, Racker E (1962) Formaldehyde-dehydrogenase from Baker's yeast. J Biol Chem 237:3279–3281
Ruhland AR, Brendel M (1979) Mutagenesis by cytostatic alkylating agents in yeast strains of differing repair capacities. Genetics 92:83–97
Ruhland AR, Haase E, Siede W, Brendel M (1981) Isolation of yeast mutants sensitive to the bifunctional alkylating agent nitrogen mustard. Mol Gen Genet 181:346–351
Ruhland AR, Brendel M, Haynes RH (1986) Hyperresistance to DNA damaging agents in yeast. Curr Genet 11:211–215
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual. Cold Spring Harbour Laboratory Press, Cold Spring Harbour, New York
Sanger F, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Sikorski RS, Hieter P (1989) A system of shuttle vectors and yeast host strains designed for efficient manipulation of DNA in Saccharomyces cerevisiae. Genetics 122:19–27
Tautz D, Renz M (1983) An optimized freeze-squeeze method for recovering of DNA fragments from agarose gels. Anal Biochem 132:114–119
Uotila L, Koivusalo M (1974) Formaldehyde dehydrogenase from human liver. J Biol Chem 249:7653–7663
Uotila L, Koivusalo M (1989) In: Dolphin D. (ed) Coenzymes and cofactors: Glutathione. Chemical, biochemical and medical aspects, vol 3, part A. Wiley, New York, pp 767–804
Vallee BL, Bazzone BJ (1983) In: Rattazzi MC (ed) Isoenzymes: current topics in biological and medical research, vol 8. Liss, New York, pp 219–244
Wagner FW, Pares X, Holmquist B, Vallee BL (1984) Physical and enzymatic properties of a class III isozyme of human liver alcohol dehydrogenase: x-ADH. Biochemistry 23:2193–2199
Wierenga RK, Terpstra P, Hol WGJ (1986) Prediction of occurrence of the ADP-binding βαβ-fold in proteins, using an amino acid sequence fingerprint. J Mol Biol 187:101–107
Zamenhoff S (1957) Preparation and assay of deoxyribonucleic acids from animal tissue. Methods Enzymol 3:696–704
Author information
Authors and Affiliations
Additional information
Communicated by W. Gajewski
Rights and permissions
About this article
Cite this article
Weimer, E.P., Rao, E. & Brendel, M. Molecular structure and genetic regulation of SFA, a gene responsible for resistance to formaldehyde in Saccharomyces cerevisiae, and characterization of its protein product. Molec. Gen. Genet. 237, 351–358 (1993). https://doi.org/10.1007/BF00279438
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00279438