Abstract
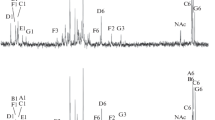
Polysaccharides excreted by cowpea Rhizobium strains JLn(c) and RA-1 were mixtures of complex acidic exopolysaccharides and low molecular weight neutral glucans. These polymers were fractionated using gel filtration chromatography. Purified fractions of the acidic heteropolymer reacted with peanut agglutinin to give precipitin bands when subjected to Ouchterlony gel diffusion. The acidic exopolysaccharide was found to contain mainly glucose, galactose, glucuronic acid, mannose and fucose. The non-carbohydrate substituents of the acidic heteropolymer were pyruvate, acetate and uronate which were identified by infrared and proton nuclear magnetic resonance spectroscopy as well as by chemical analysis.
Similar content being viewed by others
Abbreviations
- EPS:
-
Extracellular polysaccharide
- YEM:
-
yeast extract mannitol
- PNA:
-
peanut agglutination
- 1H-NMR:
-
proton nuclear magnetic resonance
References
ApteR, ModiVV (1983) A receptor protein from a host plant, Trigonella foenum-graecum, which binds to Rhizobium meliloti strains. Experientia 39:509–510
AshwellG (1966) Complex carbohydrates. In: ColowickSP, KaplanNO (eds) Methods in enzymology, vol 8. Academic Press, New York, pp 85
BohloolBB, SchmidtEL (1974) Lectins: a possible basis for specificity in the Rhizobium-legume root nodule symbiosis. Science 185:269–271
CarlsonRW, HanleyB, RolfeBG, DjordejevicMA (1986) A structural comparison of the acidic extracellular polysaccharides from Rhizobium trifolii mutants affected in root hair infection. Plant Physiol 80:134–137
ChakravortyAK, ZurkowskiW, ShineJ, RolfeBG (1982) Symbiotic nitrogen fixation: molecular cloning of Rhizobium genes involved in exopolysaccharide synthesis and effective nodulation. J Mol Appl Genet 1:585–596
ChenH, BatleyM, RedmondJ, RolfeBG (1985) Alteration of the effective nodulation properties of a fast-growing broad host range Rhizobium due to changes in exopolysaccharide synthesis. J Plant Physiol 120:331–349
DandekarAM, ModiVV (1978) Interaction between Rhizobium japonicum phage M-1 and its receptor. Can J Microbiol 24:685–688
DischeZ (1947) A new specific color reaction of hexuronic acids. J Biol Chem 167:189–198
DudmanWF (1976) The extracellular polysaccharide of Rhizobium japonicum Compositional studies. Carbohyd Res 46:97–110
DudmanWF (1978) Structural studies of the extracellular polysaccharides of Rhizobium japonicum strains 71A, CC708 and CB 1798. Carbohyd Res 66:9–23
DudmanWF, HeidelbergerWF (1969) Immunochemistry of newly found-substituents of polysaccharides of Rhizobium species. Science 164:954–955
FinanTM, HirschAM, LeighJA, JohansenE, KuldauGA, DeeganS, WalkerGC, SignerER (1985) Symbiotic mutants of Rhizobium meliloti that uncouple plant from bacterial differentiation. Cell 40:869–877
FriedmanBA, DuganPR, PfisterRM, RemsenCC (1968) Fine structure and composition of the zoological matrix surrounding Zoogloea vamigera. J Bacteriol 96:2144–2153
GardiolAE, HollingsworthRI, DazzoFB (1987) Alteration of surface properties in a Tn 5 mutant strain of Rhizobium trifolii 0403. J Bacteriol 169:1161–1167
GhaiSK, HimasatsuM, AmenuraA, HaradT (1980) Production and chemical composition of extracellular polysaccharides of Rhizobium. J Gen Microbiol 122:33–40
HestrinS (1949) The reaction of acetyl choline and other carboxylic acid derivatives with hydroxylamine and its analytical application. J Biol Chem 180:249–261
HollingsworthR, SmithE, AhmadMR (1985) Chemical composition of extracellular polysaccharide of cowpea rhizobia. Arch Microbiol 142:18–20
HollingsworthRI, DazzoFB, HallengaK, MuselmanB (1988) The complete structure of the trifoliin A lectin—binding capsular polysaccharide of Rhizobium trifolii 843. Carbohydr Res 172:97–112
KatoG, MaruyamaY, NakamuraM (1981) Involvement of lectins in Rhizobium pea recognition. Plant Cell Physiol 66:609–614
KennedyLD, BaileyRW (1976) Monomethyl sugars in extracellular polysaccharides from slow growing rhizobia. Carbohydr Res 49:451–454
KijneJW, SmitG, DiazCL, LugtenbergJJ (1988) Lectin-enhanced accumulation of manganese-limited Rhizobium leguminosarum cells on pea root hair tips. J Bacteriol 170:2994–3000
LeighJA, LeeCC (1988) Characterization of polysaccharides of Rhizobium meliloti exo mutants that form ineffective nodules. J Bacteriol 170:3327–3332
LeighJA, SignerER, WalkerGC (1985) Exopolysaccharide-deficient mutants of Rhizobium melilotti that form ineffective nodules. Proc Natl Acad Sci USA 83:6231–6235
LeighJA, ReedJW, HanksJF, HirschAM, WalkerGC (1987) Rhizobium meliloti mutants that fail to succinylate their calcoflour—binding exopolysaccharide are defective in nodule invasion. Cell 51:579–587
MariaH, ZevenhuizenLPTM (1971) Formation of cellulose fibrils by Gram-negative bacteria and their role in bacterial flocculation. Arch Mikrobiol 78:42–57
MillerGL (1959) Use of dinitrosalicyclic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
ModiM, ShahKS, ModiVV (1985) Isolation and characterization of catechol-like siderophore from cowpea Rhizobium RA-1. Arch Microbiol 141:156–158
ModyBR, ModiVV (1987) Peanut agglutinin induced alterations in capsular and extracellular polysaccharide synthesis and explanta nitrogenase activity of cowpea rhizobia. J Biosci 12:289–296
MortAJ, BauerWD (1980) Composition of the capsular and extracellular polysaccharides of Rhizobium japonicum. Plant Physiol 66:158–163
MüllerP, HynesM, KappD, NiehausK, PühlerA (1988) Two classes of Rhizobium meliloti infection mutants differ in exopolysaccharide production and in coinoculation properties with nodulation mutants. Mol Gen Genet 211:17–26
PaauAS, LepsWT, BrillWJ (1981) Agglutinin from alfalfa necessary for binding and nodulation by Rhizobium meliloti. Science 213:1513–1515
SloenkerJH, OrentasDG (1962) Pyruvic acid, a unique component of an exocellular bacterial polysaccharide. Nature (Lond) 194:478–479
SommeR (1974) Chemical analysis of exocellular, acid polysaccharides from seven Rhizobium strains. Carbohyd Res 33:89–96
StaceyG, PaauAS, BrillWJK (1980) Host recognition in Rhizobium soybean symbiosis. Plant Physiol 66:609–614
SutherlandIW (1986) Industrially useful microbial polysaccharides. Microbiol Sci 3:5–9
TsienHC, SchmidtEL (1977) Polarity in the exponential-phase of Rhizobium japonicum cells. Can J Microbiol 23:1274–1284
VincentJM (1970) A manual for the practical study of the root nodule bacteria. Blackwell Scientific Publications; Oxford
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mody, B., Bindra, M. & Modi, V. Extracellular polysaccharides of cowpea rhizobia: compositional and functional studies. Arch. Microbiol. 153, 38–42 (1989). https://doi.org/10.1007/BF00277538
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00277538