Summary
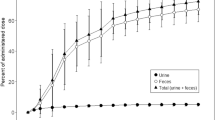
The pharmacokinetics of ifosfamide (I) were determined in ten patients with bronchogenic carcinoma. In seven patients, doses of 1 and 2 g (I) were given both as a bolus orally and later intravenously and were well tolerated. A further three patients received 5 g (I) as a single oral dose but in two this produced reversible CNS toxicity and severe vomiting. The area under the curve (AUC, μg.h.l-1) for the 1-g dose was the same following oral and i. v. treatment and this was also true for the 2-g doses. There was a proportionate increase in the AUC for the 5-g oral dose, indicating 100% bioavailability at all three dose levels. We conclude that doses up to 2 g by mouth represent a well-tolerated alternative route of administration.
Similar content being viewed by others
References
Brade WP, Herdrich K, Varini M (1985) Ifosfamide — pharmacology, safety and therapeutic potential. Cancer Treat Rev 12: 1–47
Brock N (1980) Konzeption und Wirkmechanismus von Uromitexan (Mesna) Beitr Onkol 5 1–12
Craeven PJ, Allen LM, Cohen MH, Nelson RL (1976) Studies on the clinical pharmacology and toxicology of ifosfamide (NSC 109724). Cancer Treat Rep 60 445–449
Goldin A, Venditti JM, Kline I, Wodinsky I, Little AD, Schabel FM (1977) Preclinical investigations with ifosfamide in relation to cyclophosphamide. Proc Int Holoxan Symposium, D∭seldorf 19–28
Goldin A (1982) Ifosfamide in experimental systems. Semin Oncol 9 [Suppl 1] 14–23
Karnofsky DA, Abelmann WH, Craver LF, Burshenal JH (1948) The use of the nitrogen mustards in the palliative treatment of carcinoma. Cancer 1 634–656
Klein H, Dias P, Christian E, Coerper C (1984) Therapeutic effects of single-push or fractionated injections or continous infusion of oxazaphosphorine (cyclophosphamide, ifosfamide, ASTA Z 7557). Cancer 54 1193–1203
Margison JM, Cerny T, Thatcher N, Wilkinson PM (1986) A simple quantitative HPLC assay for ifosfamide in biological fluids. Bromed Chromat 1 (3) (in press)
McNiel NO and Morgan LR (1981) The bioavailability of oral and intravenous ifosfamide in the treatment of bronchogenic carcinoma. Int J Clin Pharmacol Toxicol 19: 490–493
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47 207–214
Morgan LR, Harrison EF, Hawke JE, Hunter HL, Costanzi JJ, Plotkin D, Tucker WG, Worrall PM (1982) Toxicity of single- v. s. fractionated-dose Ifosfamide in non-small cell lung cancer a multi-center study. Semin Oncol 9 (Suppl. 1) 66–70
Seeber S, Niederle N, Osieka N, Osieka R, Schuette J (1984) Experimentelle und klinische Untersuchungen zur Wirksamkeit von Ifosfamide bei refrakt→en Neoplasien. Tumor Diagnost Ther 5 39–43
Wilkinson PM, O'Neill PA, Thatcher N, Lucas SB (1983) Pharmacokinetics of high-dose cyclophosphamide in patients with metastatic bronchogenic carcinoma. Cancer Chemother Pharmacol 11 196–199
Author information
Authors and Affiliations
Additional information
T. Cerny is the recipient of an EORTC Research Fellowship
Rights and permissions
About this article
Cite this article
Cerny, T., Margison, J.M., Thatcher, N. et al. Bioavailability of ifosfamide in patients with bronchial carcinoma. Cancer Chemother. Pharmacol. 18, 261–264 (1986). https://doi.org/10.1007/BF00273399
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00273399