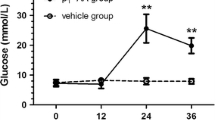
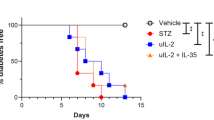
Summary
Anti-B cell auto-immunity may play a role in the pathogenesis of diabetes in mice resulting from multiple subdiabetogenic doses of the pancreatic B cell toxin, streptozotocin. In the present study we have investigated the cytotoxic anti-B cell response in these mice. A major role for B lymphocytes, macrophages, or their products in the cytotoxic response originally detected in vitro was eliminated by passing splenocytes from the mice treated with multiple subdiabetogenic doses of streptozotocin over a nylon wool column. The removal of the adherent cells enhanced the cytotoxicity against a rat insulinoma cell line in vitro by that expected due to enrichment of T-lymphocytes by approximately twofold. The induction of diabetes after multiple subdiabetogenic doses of streptozotocin is strain dependent. Mice of five strains were immunized with rat insulinoma cells, but only splenocytes from the two strains susceptible to multiple subdiabetogenic doses of streptozotocin demonstrated a significant cytotoxic response against the rat insulinoma cells in vitro. Mice pre-immunized with either the rat insulinoma cells or with syngeneic islets labelled in vitro with the hapten trinitrophenol developed hyperglycaemia more rapidly than control mice after multiple subdiabetogenic doses of streptozotocin. In the latter experiment the control mice immunized with complete Freund's adjuvant alone also became hyperglycaemic after a modified multiple subdiabetogenic dose of streptozotocin that did not cause diabetes in non-immunized mice. In mice pre-treated with either adjuvant or cyclophosphamide and then given a modified multiple subdiabetogenic dose of streptozotocin (35 mg/kg × 5 rather than 40 mg/ kg) the degree of hyperglycaemia was reduced and there was no protective effect of cyclophosphamide. However, the mice pre-treated with adjuvant again developed hyperglycaemia more rapidly and to a much higher level than did the mice given multiple subdiabetogenic doses of streptozotocin only. These additional data further support the hypothesis that B-cell destruction after multiple subdiabetogenic doses of streptozotocin results from triggering of an immune response against these insulin-producing cells.
Article PDF
Similar content being viewed by others
References
Like AA, Rossini AA (1976) Streptozotocin-induced pancreatic insulitis. A new model of diabetes mellitus. Science 193: 415–417
Rossini AA, Like AA, Appel MC, Chick WL, Cahill GF Jr (1977) Studies of streptozotocin-induced insulitis and diabetes. Proc Nat Acad Sci (USA) 74: 2485–2489
Rossini AA, Williams RM, Appel MC, Like AA (1978) Complete protection from streptozotocin-induced diabetes in mice. Nature 276: 182–184
Buschard K, Rygaard J (1978) T-lymphocytes transfer streptozotocin-induced diabetes mellitus in mice. Acta Pathol Microbiol Scand [C] 86: 277–282
Beattie G, Lannum R, Lipstick J, Kaplan NO, Osler AG (1980) Streptozotocin-induced diabetes in athymic and conventional BALB/c mice. Diabetes 29: 146–150
Nakamura M, Nagafuchi S, Yamaguchi K, Takaki R (1984) The role of thymic immunity and insulitis in the development of streptozotocin-induced diabetes in mice. Diabetes 33: 894–900
Paik S-G, Blue ML, Fleischer N, Shen SI (1982) Diabetes susceptibility of BALB/cBOM mice treated with streptozotocin: inhibition by lethal irradiation and restoration by splenec lymphocytes. Diabetes 31: 808–815
Hahn HJ, Barnstorf K, Nadrowitz R, Besch W, Schmidt W (1982) The effect of radioimmunosuppression on the development of low dose streptozotocin diabetes. Acta Biol Med Ger 41: 1191–1197
Nedergaard M, Egeberg J, Kromann H (1983) Irradiation protects against pancreatic islet degeneration and hyperglycemia following streptozotocin treatment of mice. Diabetologia 24: 82–86
Blue ML, Shin S-I (1984) Diabetes induced by subdiabetogenic doses of streptozotocin in BALB/cBOM mice: Noninvolvement of host B lymphocyte function. Diabetes 33: 105–113
Kolb H, Kiesel U (1981) Localization of genes controlling an experimental model of human IDDM within and without of the major histocompatibility complex. Immunobiology 160: 58–62
Kiesel U, Kolb H (1982) Low-dose, streptozotocin-induced diabetes is under the genetic control of the major histocompatibility complex in mice. Diabetologia 23: 69–71
Kiesel U, Falkenberg FW, Kolb H (1983) Genetic control of low-dose streptozotocin-induced autoimmune diabetes in mice. J Immunol 130: 1719–1722
Wolf J, Lilly F, Shin S-I (1984) The influence of genetic background on the susceptibility of inbred mice to streptozotocin-induced diabetes. Diabetes 33: 567–571
Leiter EH (1984) Genetic control of pathogenesis of diabetes in C3H mice: Influence of the major histocompatibility complex. Diabetes 33: 1068–1072
Kim YT, Steinberg C (1984) Immunological studies on the induction of diabetes in experimental mice. Diabetes 33: 771–777
Koevary S, Rossini AA, Stoller W, Chick W, Williams RM (1983) Passive transfer of diabetes in the BB rat. Science 220: 727–729
Laupacis A, Gardell C, Dupre J, Stiller CR, Keown P, Wallace AC, Thibert P (1983) Cyclosporin prevents diabetes in BB Wistar rats. Lancet 1: 10–12
Like AA, Dirodi V, Thomas S, Guberski DL, Rossini AA (1984) Prevention of diabetes mellitus in the BB/W rat with Cyclosporin A. Am J Pathol 117: 92–97
Sestier C, Odent-Pogu S, Bonneville M, Maurel C, Lang F, Sai P (1985) Cyclosporin enhances diabetes induced by low dose streptozotocin treatment in mice. Immunol Lett 10: 57–60
Andersson A, Borg H, Hallberg A, Hellerström C, Sandler S, Schnell A (1984) Direct toxicity of Cyclosporin on beta cells in tissue culture. Diabetologia 27: 66–69
Bonnevie-Neilsen V, Steffes MW, Lernmark A (1981) A major loss in islet cell mass and B cell function precedes hyperglycemia in mice given multiple low doses of streptozotocin. Diabetes 30: 424–429
Cossel L, Schneider E, Kuttler B, Schmidt S, Wohlrab F, Schade J, Bochmann Ch (1985) Low dose streptozotocin-induced diabetes in mice. Metabolic, light microscopic, histochemical, immunofluorescence microscopic, electron microscopic and morphometric findings. Exp Clin Endocrinol 85: 7–26
Schein PS, Loftus S (1968) Streptozotocin: depression of mouse liver pyridine nucleotides. Cancer Res 28: 1501–1506
Robbins MJ, Sharp RA, Slonim AE, Burr IM (1980) Protection against streptozotocin-induced diabetes by superoxide dismutase. Diabetologia 18: 55–58
Sandler S, Andersson A (1985) Modulation of streptozotocin-induced insulitis and hyperglycemia in the mouse. Acta Pathol Microbiol Immunol Scand (A) 93: 93–98
Gold G, Manning M, Heldt A, Nowlain R, Pettit JR, Grodsky GM (1981) Diabetes induced by multiple subdiabetogenic doses of streptozotocin. Lack of protection by exogenous superoxide dismutase. Diabetes 30: 634–638
McEvoy RC, Andersson J, Sandler S, Hellerström C (1984) Multiple, lowdose streptozotocin-induced diabetes in the mouse. Evidence for stimulation of a cytotoxic, cellular immune response against an insulin-producing beta cell line. J Clin Invest 74: 715–722
McKay P, Boulton A, Rabinovitch A (1985) Lymphoid cells of BB/W rats are cytotoxic to islet beta cells in vitro. Diabetes 34: 706–709
McDevitt HO (1982) Regulation of the immune response by the major histocompatibility system. N Engl J Med 303: 1514–1517
Marrack P, Kappler J (1986) The T cell and its receptor. Sci Am 254: 36–45.
McEvoy RC, Franklin BH, Ginsberg-Fellner F (1986) Biological differences in a rat insulinoma cell line obtained from different laboratories do not affect binding of human anti-islet immunoglobulins. Diabetes Res 3: 339–344
Gazdar AF, Chick WL, Oie HK, Sims HL, King DL, Weir GC, Lauris V (1980) Continuous, clonal insulin- and somatostatin-secreting cell line established from a transplantable rat islet cell tumor. Proc Nat Acad Sci (USA) 77: 3319–3323
Bloom BR, David JR (1973) A rapid method for the isolation of functional thymus-derived murine lymphocytes. Eur J Immunol 3: 645–649
Lacy PE, Kostianovsky M (1967) Method for the isolation of intact islets of Langerhans from rat pancreas. Diabetes 16: 35–39
Rollinghoff M, Starzinski-Powitz A, Pfizenmaier K, Wagner H (1977) Cyclophosphamide-sensitive T-lymphocytes suppress the in vivo generation of antigen-specific cytotoxic T-lymphocytes. J Exp Med 145: 455–459
Sandler S, Andersson A (1981) Islet implantation into diabetic mice with pancreatic insulitis. Acta Pathol Microbiol Immunol Scand [A] 89: 107–112
Raabo E, Terkildsen TC (1960) On the enzymatic determination of blood glucose. Scand J Clin Lab Invest 12: 402–404
Kiesel U, Kolb H (1983) Suppressive effect of antibodies to immune response gene products on the development of low dose streptozotocin induced diabetes. Diabetes 32: 869–871
Mandrup-Poulsen T, Bendtzen K, Nerup J, Dinarello CA, Svenson M, Nielsen JH (1986) Affinity-purified human Interleukin-I is cytotoxic to isolated islets of Langerhans. Diabetologia 29: 63–67
Ziegler M, Ziegler B, Hehme B, Deitz H, Hildmann W, Kauert C (1984) Autoimmune destruction of pancreatic beta cells in rats induced by combination treatment with a low dose of streptozotocin and complete Freund's adjuvant. Biomed Biochim Acta 43: 675–681
Bottazzo GF, Dean BM, McNally JM, MacKay EH, Swift PGF, Gamble DR (1985) In situ characterization of autoimmune phenomena and expression of HLA molecules in the pancreas in diabetic insulitis. N Engl J Med 313: 353–359
Dean BM, Walker R, Bone AJ, Baird JD, Cooke A (1985) Prediabetes in the spontaneously diabetic BB/E rat: lymphocytes subpopulations in the pancreatic infiltrate and expression of rat MHC class II molecules in the endocrine cells. Diabetologia 28: 464–466
Ulrichs K, Müller-Ruchholtz W (1985) MHC Class II antigen expression on various cells of normal and activated pancreatic islets. Diag Immunol 3: 47–55
Campbell IL, Wong GHW, Schrader JW, Harrison LC (1985) Interferon-gamma enhances the expression of the major histocompatibility Class I antigens on mouse pancreatic beta cells. Diabetes 34: 1205–1209
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McEvoy, R.C., Thomas, N.M., Hellerström, C. et al. Multiple low-dose streptozotocin-induced diabetes in the mouse: further evidence for involvement of an anti-B cell cytotoxic cellular auto-immune response. Diabetologia 30, 232–238 (1987). https://doi.org/10.1007/BF00270421
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00270421