Summary
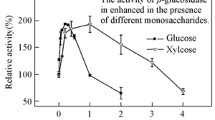
Clostridium stercorarium cultures grown on cellobiose contain both an extracellular and a cell-bound β-glucosidase activity. A substantial portion of the cell-bound enzyme could be extracted by osmotic shock, suggesting a periplasmic localization. The β-glucosidase present in culture supernatants was purified to homogeneity. It was found to be identical in all aspects tested with the cell-bound β-glucosidase. The enzyme exists as a monomer with an apparent molecular weight of 85.000 (SDS-PAGE) and a pI of 4.8. It shows optimal activity as pH 5.5 and 65° C. Thiol groups are essential for enzyme activity. In the presence of reducing agents and divalent cations the half-life of the purified enzyme was more than 5 h at 60°C. The enzyme hydrolyses at different rates a wide range of substrates including aryl-β-glucosides, cellobiose, and disordered cellulose. K m values were determined as 0.8 mM for p-nitrophenyl-β-glucoside (PNPG) and 33 mM for cellobiose. The cellular localization and the substrate specificity pattern are consistent with a dual role of the C. stercorarium β-glucosidase in cellulose saccharification: (1) Cleavage of cellobiose formed by exoglucanase and (2) degradation of cellodextrins produced by endoglucanase action.
Similar content being viewed by others
References
Aiba S, Kitai K, Imanaka T (1983) Cloning of a thermostable alpha-amylase gene from Bacillus stearothermophilus in Bacillus stearothermophilus and Bacillus subtilis. Appl Environ Microbiol 46:1059–1065
Ait N, Creuzet N, Cattaneo J (1979) Characterization and purification of thermostable β-glucosidase from Clostridium thermocellum. Biochem Biophys Res Commun 90:537–546
Ait N, Creuzet N, Cattaneo J (1982) Properties of β-glucosidase purified from Clostridium thermocellum. J Gen Microbiol 128:569–577
Alexander JK (1968) Purification and specificity of cellobiose phosphorylase from Clostridium thermocellum. J Biol Chem 243:2899–2904
Barras F, Chambost JP, Chippaux M (1984) Cellobiose metabolism in Erwinia: A genetic study. Mol Gen Genet 197:490–496
Breuil C, Wojtczak G, Saddler JN (1986) Production and localization of cellulases and β-glucosidase from the thermophilic fungus Thielavia terrestris. Biotechnol Lett 8:673–676
Bronnenmeier K, Staudenbauer WL (1988) Resolution of Clostridium stercorarium cellulase by fast protein liquid chromatography (FPLC). Appl Microbiol Biotechnol 27:432–436
Coughlan MP (1985) The properties of fungal and bacterial cellulases with comment on their production and application. Biotechnol Genet Eng Rev 3:39–109
Creuzet N, Frixon C (1983) Purification and characterization of an endoglucanase from a newly isolated thermophilic anaerobic bacterium. Biochimie 65:149–156
Hägerdal B, Ferchack JD, Pye EK (1980) Saccharification of cellulose by the cellulolytic enzyme system of Thermomonospora sp. Biotechnol Bioeng 22:1515–1526
Johnson EA, Madia A, Demain AL (1981) Chemically defined minimal medium for growth of the anaerobic cellulolytic thermophile Clostridium thermocellum. Appl Environ Microbiol 41:1060–1062
Johnson EA, Sakajoh M, Halliwell G, Madia A, Demain AL (1982) Saccharification of complex cellulosic substrates by the cellulase system from Clostridium thermocellum. Appl Environ Microbiol 43:1125–1132
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Madden RH (1983) Isolation and characterization of Clostridium stercorarium sp. nov., cellulolytic thermophile. Int J Syst Bacteriol 33:837–840
Nanda M, Bisaria VS, Ghose TK (1982) Localization and release mechanism of cellulases in Trichoderma reesei QM 9414. Biotechnol Lett 4:633–638
Ohmiya K, Shiria M, Kurachi Y, Shimizu S (1985) Isolation and properties of β-glucosidase from Ruminococcus albus. J Bacteriol 161:432–434
Patchett ML, Daniel RM, Morgan HW (1987) Purification and properties of a stable β-glucosidase from an extremely thermophilic anaerobic bacterium. Biochem J 243:779–787
Schimz KL, Broll B, John B (1983) Cellobiose phosphorylase (EC 2.4.1.20) of Cellulomonas: occurrence, induction, and its role in cellobiose metabolism. Arch Microbiol 135:241–249
Schmid G, Wandrey C (1987) Purification and partial characterization of a cellodextrin glucohydrolase (β-glucosidase) from Trichoderma reesei strain QM 9414. Biotechnol Bioeng 30:571–585
Schwarz WH, Bronnenmeier K, Gräbnitz F, Staudenbauer WL (1987) Activity staining of cellulases in polyacrylamide gels containing mixed linkage β-glucans. Anal Biochem 164:72–77
Sedmark JJ, Grossberg SE (1977) A rapid, sensitive assay for protein using Coomassie brilliant blue G250. Anal Biochem 79:544–552
Sheth K, Alexander JK (1969) Purification and properties of β-1,4-oligoglucan: orthophosphate glucosyltransferase from Clostridium thermocellum. J Biol Chem 244:457–464
Stoppok W, Rapp P, Wagner F (1982) Formation, location, and regulation of endo-1,4-β-glucanases and β-glucosidases from Cellulomonas uda. Appl Environ Microbiol 44:44–53
Tsukagoshi N, Ihara H, Yamagata H, Udaka S (1984) Cloning and expression of a thermostable α-amylase gene from Bacillus stearothermophilus in Escherichia coli. Mol Gen Genet 193:58–63
Wood TM (1971) The cellulases of Fusarium solani. Purification and specificity of the β-1,4-glucanase and β-glucosidase components. Biochem J 121:353–362
Wood TM, McCrae SI (1982) Purification and some properties of a (1→4)-β-d-glucan glucohydrolase associated with the cellulase from the fungus Penicillium funiculosum. Carbohydrate Res 110:291–303
Woodward J, Wiseman A (1982) Fungal and other β-glucosidases—their properties and applications. Enzyme Microb Technol 4:73–79
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bronnenmeier, K., Staudenbauer, W.L. Purification and properties of an extracellular β-glucosidase from the cellulolytic thermophile Clostridium stercorarium . Appl Microbiol Biotechnol 28, 380–386 (1988). https://doi.org/10.1007/BF00268200
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00268200