Summary
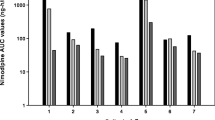
Nimodipine pharmacokinetics was investigated in 12 Chinese patients with acute subarachnoid haemorrhage receiving an IV infusion of 1.6 or 2 mg/h (based on estimated body weight) for 10 days. Peripheral venous blood samples were collected for up to 4 days and plasma nimodipine was assayed by GC/ECD. The mean value was taken as the steady state concentration (Css) and Clearance (CL) (hourly dose/Css) was calculated. Eight survivors were given oral nimodipine (60 or 90 mg) every 6h (based on body weight), blood was sampled over 6 h and the plasma nimodipine level determined. The values for Css, CL and CL·kg−1 were 33.5 μg·l−1, 58 l·h−1 and 1.0 l·h−1·kg−1 respectively; in survivors receiving the drug orally, bioavailability of the 30 mg tablet was 9%. In one very sick patient given crushed tablets by naso-gastric tube, the AUC was very low; in vitro studies indicated that adsorption of nimodipine by the tubing was unlikely to have been the cause.
The pharmacokinetic findings in Chinese patients are comparable to previously reported values in Caucasians.
Similar content being viewed by others
References
Hawes CW, Gourtley JK, Heistad DD (1983) Effects of nimodipine cerebral on blood flow. J Pharmacol exp Ther 225: 22–28
Langley SL, Sorkin EM (1989) Nimodipine. A Review of its pharmacodynamic and pharmacokinetic properties, and therapeutic potential in cerebrovascular disease. Drugs 37: 669–699
Rämsch K-D, Graefe K-H, Scherling D, Sommer J, Zielger R (1986) Pharmacokinetics and metabolism of calcium-blocking agents nifedipine, nitrendipine and nimodipine. Am J Nephrol 6 [Suppl 1]: 73–80
Barr JA, Goodnight JH, Sall JP, Helwig JT (1979) A Users Guide to SAS, SAS Institute, Rayleigh, North Carolina, USA
Rämsch K-D, Ahr G, Tettenborn D, Auer LM (1985) Overview on pharmacokinetics of nimodipine in healthy volunteers and in patients with subarachnoid hemorrhage. Neurochirurgia 28: 74–78
Vinge E, Andersson KE, Brandt L, Ljunggren B, Nilsson LG, Rosendal-Helgesen S (1986) Pharmacokinetics of nimodipine in patients with aneurysmal subarachnoid haemorrhage. Eur J Clin Pharmacol 30: 421–425
Kirch W, Rämsch KD, Dührsen U, Ohnhaus EE (1984) Clinical pharmacokinetics of nimodipine in normal and impaired renal function. Int J Clin Pharm Res IV: 381–384
Auer LM, Ito Z, Suzuki A, Ohta H (1982) Prevention of sympathic vasospasm by topically applied nimodipine. Acta Neurochir 63: 297
Meyer H, Wehinger E, Bossert F, Scherling D (1983) Nimodipine synthesis and metabolic pathways. Drug Res 339: 106–112
Laursen J, Jensen F, Mikkelsen E, Jakobsen (1988) Nimodipine treatment of subarachnoid hemorrhage. Clin Neurol Neurosurg 90: 329–337
Abernathy DR, Greenblatt DJ, Divoll M, Harmatz JS, Shader RI (1980) Alterations in drug distribution and clearance due to obesity. J Pharmacol Exp Ther 217: 681
Kumana CR, Lauder IJ, Chan M, Ko W, Lin HJ (1987) Differences in diazepam pharmacokinetics in Chinese and White Caucasians — Relation to body lipid stores. Eur J Clin Pharmacol 32: 211–215
Tolosa E, Soler-Singla L, Segura J, Ribera G, Esteve MV, Caturla MC, Graus F, Albors M, Badenas JM, Bigorra L (1989) Comparative pharmacokinetic study of several routes of administration of nimodipine in acute ischemic stroke patients. New Trends Clin Neuropharmacol 3: 10
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumana, C.R., Kou, M., Yu, Y.L. et al. Investigation of nimodipine pharmacokinetics in Chinese patients with acute subarachnoid haemorrhage. Eur J Clin Pharmacol 45, 363–366 (1993). https://doi.org/10.1007/BF00265956
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00265956